18.4 Electrode and Cell Potentials
Learning Objectives
- Determine standard cell potentials for oxidation-reduction reactions
- Use standard reduction potentials to determine the better oxidizing or reducing agent from among several possible choices
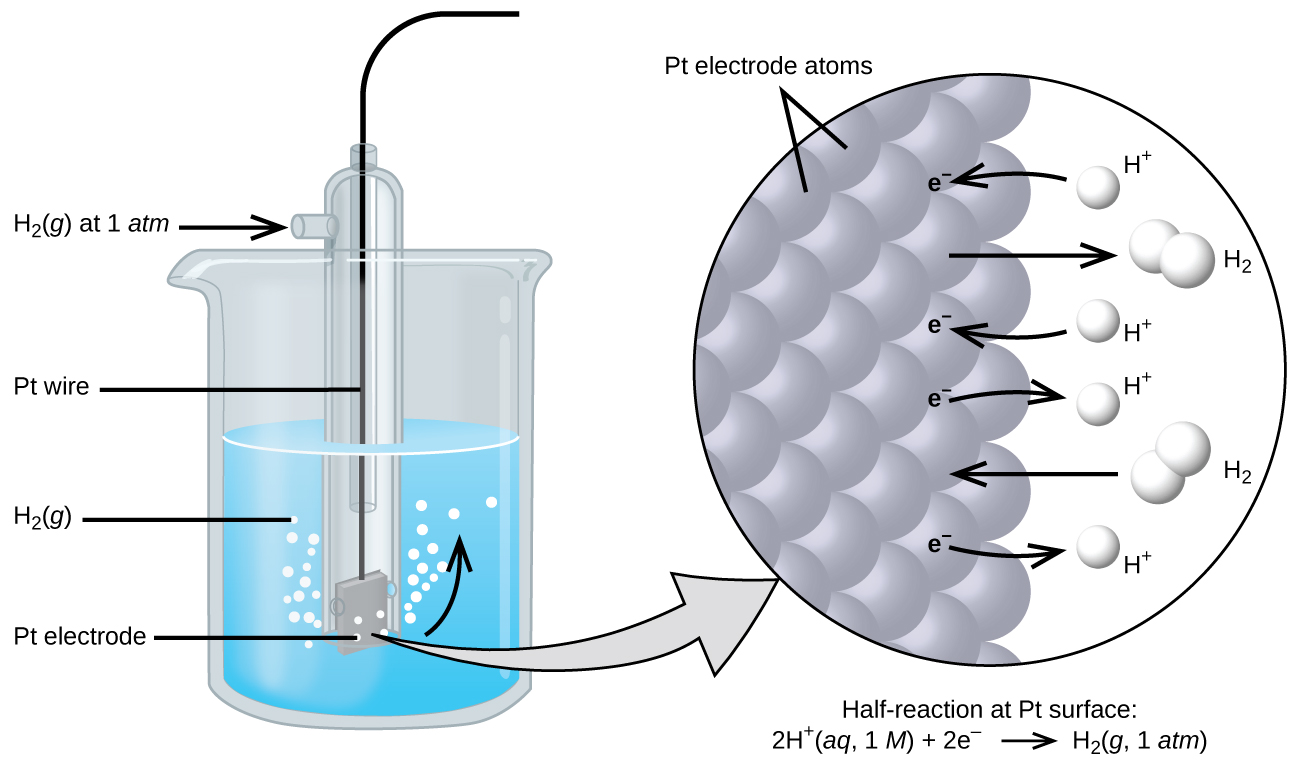
The cell potential in 18.3 – Galvanic Cells results from the difference in the electrical potentials for each electrode. While it is impossible to determine the electrical potential of a single electrode, we can assign an electrode the value of zero and then use it as a reference. The electrode chosen as the zero is shown in Figure 18.4a and is called the standard hydrogen electrode (SHE). The SHE consists of 1 atm of hydrogen gas bubbled through a 1 M HCl solution, usually at room temperature. Platinum, which is chemically inert, is used as the electrode. The reduction half-reaction chosen as the reference is
E° is the standard reduction potential. The superscript “°” on the E denotes standard conditions (1 bar or 1 atm for gases, 1 M for solutes). The voltage is defined as zero for all temperatures.
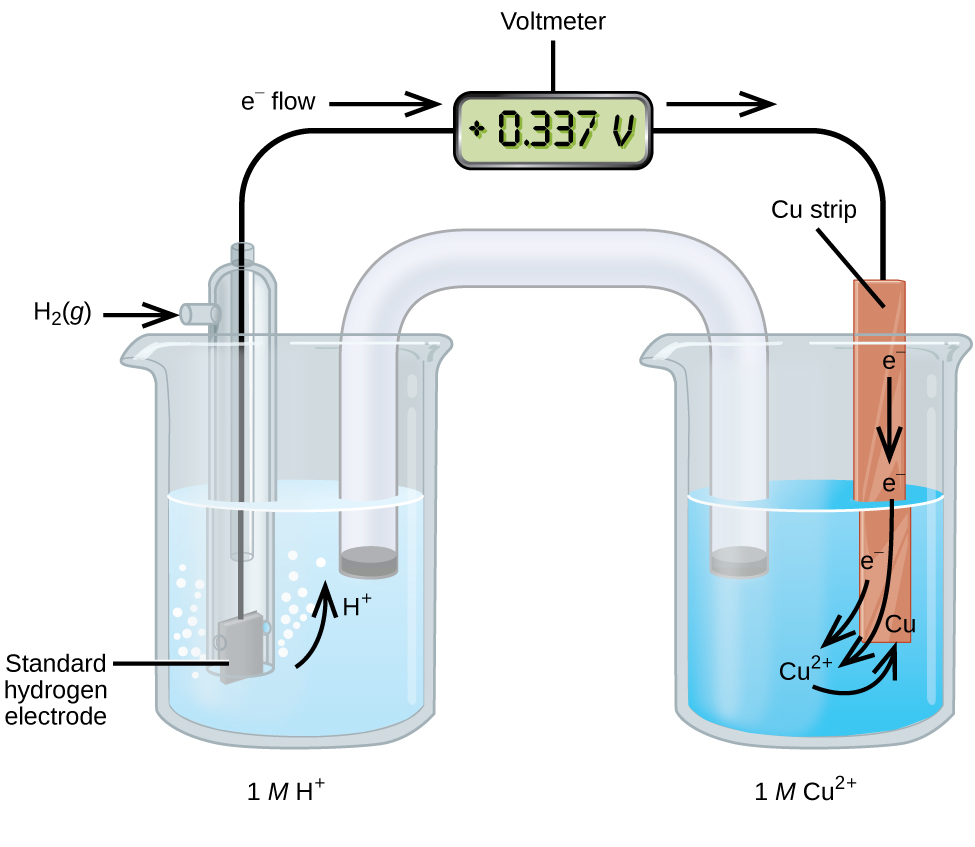
A galvanic cell consisting of a SHE and Cu2+/Cu half-cell can be used to determine the standard reduction potential for Cu2+ (Figure 18.4b). In cell notation, the reaction is
Electrons flow from the anode to the cathode. The reactions, which are reversible, are
[latex]\begin{array}{lr @{{}\longrightarrow{}} l} \text{Anode (oxidation):} & \text{H}_2(g) & \longrightarrow 2\text{ H}^{+}(aq)\;+\;2\text{ e}^{-} \\[0.5em] \text{Cathode (reduction):} & \text{Cu}^{2+}(aq)\;+\;2\text{ e}^{-} & \longrightarrow \text{Cu}(s) \\[0.5em] \hline \\[-0.25em] \text{Overall:} & \text{Cu}^{2+}(aq)\;+\;\text{H}_2(g) & \longrightarrow 2\text{ H}^{+}(aq)\;+\;\text{Cu}(s) \end{array}[/latex]
The standard reduction potential can be determined by subtracting the standard reduction potential for the reaction occurring at the anode from the standard reduction potential for the reaction occurring at the cathode. The minus sign is necessary because oxidation is the reverse of reduction.
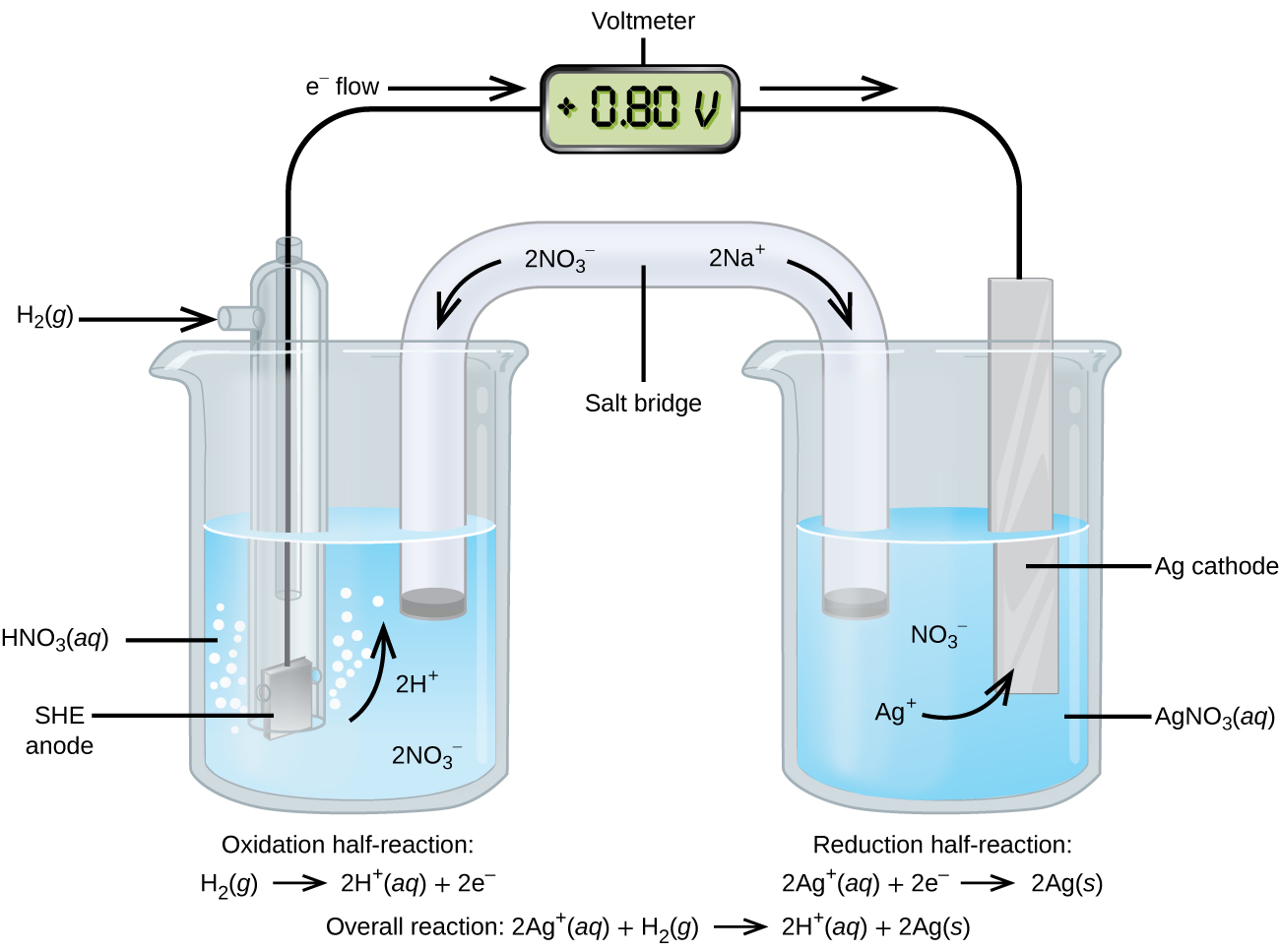
Using the SHE as a reference, other standard reduction potentials can be determined. Consider the cell shown in Figure 18.4c, where
Electrons flow from left to right, and the reactions are
The standard reduction potential can be determined by subtracting the standard reduction potential for the reaction occurring at the anode from the standard reduction potential for the reaction occurring at the cathode. The minus sign is needed because oxidation is the reverse of reduction.
It is important to note that the potential is not doubled for the cathode reaction.
The SHE is rather dangerous and rarely used in the laboratory. Its main significance is that it established the zero for standard reduction potentials. Once determined, standard reduction potentials can be used to determine the standard cell potential, [latex]E_{\text{cell}}^{\circ}[/latex], for any cell. For example, for the cell shown in Figure 18.3b in 18.3 – Galvanic Cells.
[latex]\begin{array}{lr @{{}\longrightarrow{}} l} \text{anode (oxidation):} & \text{Cu}(s) & \longrightarrow\text{Cu}^{2+}(aq)\;+\;2\text{ e}^{-} \\[0.5em] \text{cathode (reduction):} & 2\text{ Ag}^{+}(aq)\;+\;2\text{ e}^{-} & \longrightarrow 2\text{ Ag}(s) \\[0.5em] \hline \\[-0.25em] \text{overall:} & \text{Cu}(s)\;+\;2\text{ Ag}^{+}(aq) & \longrightarrow\text{Cu}^{2+}(aq)\;+\;2\text{ Ag}(s) \\[0.5em]\end{array}\\[0.5em][/latex]
Again, note that when calculating [latex]E_{\text{cell}}^{\circ}[/latex], standard reduction potentials always remain the same even when a half-reaction is multiplied by a factor. Standard reduction potentials for selected reduction reactions are shown in Table 18.4a. A more complete list is provided in Appendix M.
Tables like this make it possible to determine the standard cell potential for many oxidation-reduction reactions.
| Half-Reaction | E° (V) |
|---|---|
| F2(g) + 2 e– → 2 F–(aq) | +2.866 |
| PbO2(s) + SO42-(aq) + 4 H+(aq) + 2 e– → PbSO4(s) + 2 H2O(l) | +1.69 |
| MnO4–(aq) + 8 H+(aq) + 5 e– → Mn2+(aq) + 4 H2O(l) | +1.507 |
| Au3+(aq) + 3 e– → Au(s) | +1.498 |
| Cl2(g) + 2 e– → 2 Cl–(aq) | +1.35827 |
| O2(g) + 4 H+(aq) + 4 e– → 2 H2O(l) | +1.229 |
| Pt2+(aq) + 2 e– → Pt(s) | +1.20 |
| Br2(aq) + 2 e– → 2 Br–(aq) | +1.0873 |
| Ag+(aq) + e– → Ag(s) | +0.7996 |
| Hg22+(aq) + 2 e– → 2 Hg(l) | +0.7973 |
| Fe3+(aq) + e– → Fe2+(aq) | +0.771 |
| MnO4–(aq) + 2 H2O(l) + 3 e– → MnO2(s) + 4 OH–(aq) | +0.558 |
| I2(s) + 2 e– → 2 I–(aq) | +0.5355 |
| NiO2(s) +2 H2O(l) + 2 e– → Ni(OH)2(s) + 2 OH–(aq) | +0.49 |
| Cu2+(aq) + 2 e– → Cu(s) | +0.337 |
| Hg2Cl2(s) + 2 e– → 2 Hg(l) + 2 Cl–(aq) | +0.26808 |
| AgCl(s) + 2 e– → Ag(s) + Cl–(aq) | +0.22233 |
| Sn4+(aq) + 2 e– → Sn2+(aq) | +0.151 |
| 2 H+(aq) + 2 e– → H2(g) | 0.00 |
| Pb2+(aq) + 2 e– → Pb(s) | −0.126 |
| Sn2+(aq) + 2 e– → Sn(s) | −0.1262 |
| Ni2+(aq) + 2 e– → Ni(s) | −0.257 |
| Co2+(aq) + 2 e– → Co(s) | −0.28 |
| PbSO4(s) + 2 e– → Pb(s) + SO42-(aq) | −0.3505 |
| Cd2+(aq) + 2 e– → Cd(s) | −0.4030 |
| Fe2+(aq) + 2 e– → Fe(s) | −0.447 |
| Cr3+(aq) + 3 e– → Cr(s) | −0.744 |
| Mn2+(aq) + 2 e– → Mn(s) | −1.185 |
| Zn(OH)2(s) + 2e – → Zn(s) + 2 OH–(aq) | −1.245 |
| Zn2+(aq) + 2 e– → Zn(s) | −0.7618 |
| Al3+(aq) + 3 e– → Al(s) | −1.662 |
| Mg2+(aq) + 2 e– → Mg(s) | −2.372 |
| Na+(aq) + e– → Na(s) | −2.71 |
| Ca2+(aq) + 2 e– → Ca(s) | −2.868 |
| Ba2+(aq) + 2 e– → Ba(s) | −2.912 |
| K+(aq) + e– → K(s) | −2.931 |
| Li+(aq) + e– → Li(s) | −3.04 |
Example 18.4a
Cell Potentials from Standard Reduction Potentials
What is the standard cell potential for a galvanic cell that consists of Au3+/Au and Ni2+/Ni half-cells? Identify the oxidizing and reducing agents.
Solution
Using Table 18.4a, the reactions involved in the galvanic cell, both written as reductions, are
Galvanic cells have positive cell potentials, and all the reduction reactions are reversible. The reaction at the anode will be the half-reaction with the smaller or more negative standard reduction potential. Reversing the reaction at the anode (to show the oxidation) but not its standard reduction potential gives:
[latex]{\scriptsize \begin{array}{lr @{{}\longrightarrow{}} ll} \text{Anode (oxidation):} & \text{Ni}(s) & \longrightarrow\text{Ni}^{2+}(aq)\;+\;2\text{ e}^{-} & E_{\text{anode}}^{\circ} = E_{\text{Ni}^{2+}/\text{Ni}}^{\circ} = -0.257\;\text{V} \\[0.5em] \text{Cathode (reduction):} & \text{Au}^{3+}(aq)\;+\;3\text{ e}^{-} & \longrightarrow \text{Au}(s) & E_{\text{cathode}}^{\circ} = E_{\text{Au}^{3+}/\text{Au}}^{\circ} = +1.498\;\text{V} \end{array} \scriptsize}[/latex]
The least common factor is six, so the overall reaction is
The reduction potentials are not scaled by the stoichiometric coefficients when calculating the cell potential, and the unmodified standard reduction potentials must be used.
From the half-reactions, Ni is oxidized, so it is the reducing agent, and Au3+ is reduced, so it is the oxidizing agent.
Exercise 18.4a
A galvanic cell consists of a Mg electrode in 1 M Mg(NO3)2 solution and a Ag electrode in 1 M AgNO3 solution. Calculate the standard cell potential at 25 °C.
Check Your Answer[1]
Key Equations
- [latex]E_{\text{cell}}^{\circ} = E_{\text{cathode}}^{\circ}\;-\;E_{\text{anode}}^{\circ}[/latex]
Attribution & References
Except where otherwise noted, this page is adapted by David Wegman from “17.3 Standard Reduction Potentials” In General Chemistry 1 & 2 by Rice University, a derivative of Chemistry (Open Stax) by Paul Flowers, Klaus Theopold, Richard Langley & William R. Robinson and is licensed under CC BY 4.0. Access for free at Chemistry (OpenStax).
- [latex]{\scriptsize \text{Mg}(s)\;+\;2\text{ Ag}^{+}(aq)\;{\longrightarrow}\;\text{Mg}^{2+}(aq)\;+\;2\text{ Ag}(s)\;\;\;\;\;\;\;E_{\text{cell}}^{\circ} = 0.7996\;\text{V}\;-\;(-2.372\;\text{V}) = 3.172\;\text{V} \scriptsize}[/latex] ↵
the electrode consists of hydrogen gas bubbling through hydrochloric acid over an inert platinum electrode whose reduction at standard conditions is assigned a value of 0 V; the reference point for standard reduction potentials
the cell potential when all reactants and products are in their standard states (1 bar or 1 atm or gases; 1 M for solutes), usually at 298.15 K; can be calculated by subtracting the standard reduction potential for the half-reaction at the anode from the standard reduction potential for the half-reaction occurring at the cathode

