18.5 Batteries and Fuel Cells
Learning Objectives
- Classify batteries as primary or secondary
- List some of the characteristics and limitations of batteries
- Provide a general description of a fuel cell
A battery is an electrochemical cell or series of cells that produces an electric current. In principle, any galvanic cell could be used as a battery. An ideal battery would never run down, produce an unchanging voltage, and be capable of withstanding environmental extremes of heat and humidity. Real batteries strike a balance between ideal characteristics and practical limitations. For example, the mass of a car battery is about 18 kg or about 1% of the mass of an average car or light-duty truck. This type of battery would supply nearly unlimited energy if used in a smartphone, but would be rejected for this application because of its mass. Thus, no single battery is “best” and batteries are selected for a particular application, keeping things like the mass of the battery, its cost, reliability, and current capacity in mind. There are two basic types of batteries: primary and secondary. A few batteries of each type are described next.
Watch Dry Cell - Definition, Working Principle, History, Parts of Dry Cell, Chemical reactions (4:48 min)
Video source: TutorVista. (2010, May 5). Dry cell - Definition, working principle, history, parts of dry cell, chemical reactions [Video]. YouTube.
Primary Batteries
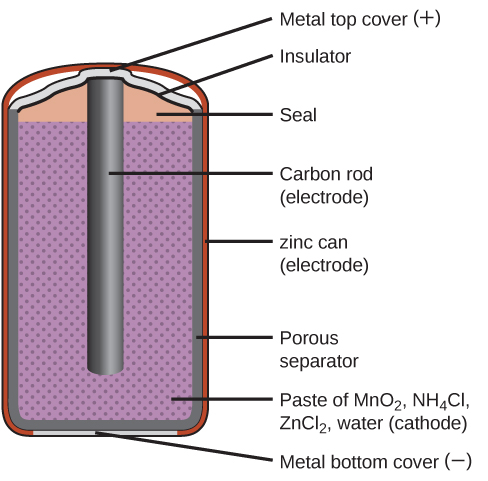
Primary batteries are single-use batteries because they cannot be recharged. A common primary battery is the dry cell (Figure 18.5a). The dry cell is a zinc-carbon battery. The zinc can serve as both a container and the negative electrode. The positive electrode is a rod made of carbon that is surrounded by a paste of manganese(IV) oxide, zinc chloride, ammonium chloride, carbon powder, and a small amount of water. The reaction at the anode can be represented as the ordinary oxidation of zinc:
The reaction at the cathode is more complicated, in part because more than one reaction occurs. The series of reactions that occurs at the cathode is approximately:
The overall reaction for the zinc–carbon battery can be represented as:
[latex]{\scriptsize 2\text{ MnO}_2(s)\;+\;2\text{ NH}_4\text{Cl}(aq)\;+\;\text{Zn}(s)\;{\longrightarrow}\;\text{Zn}^{2+}(aq)\;+\;\text{Mn}_2\text{O}_3(s)\;+\;2\text{ NH}_3(aq)\;+\;\text{H}_2\text{O}(l)\;+\;2\text{ Cl}^{-} \scriptsize}[/latex]
with an overall cell potential which is initially about 1.5 V, but decreases as the battery is used. It is important to remember that the voltage delivered by a battery is the same regardless of the size of a battery. For this reason, D, C, A, AA, and AAA batteries all have the same voltage rating. However, larger batteries can deliver more moles of electrons. As the zinc container oxidizes, its contents eventually leak out, so this type of battery should not be left in any electrical device for extended periods.
Watch Single-use cells & batteries (2:47 min)
Video source: Rees Films. (2011, September 13). Single-use cells & batteries [Video]. YouTube.
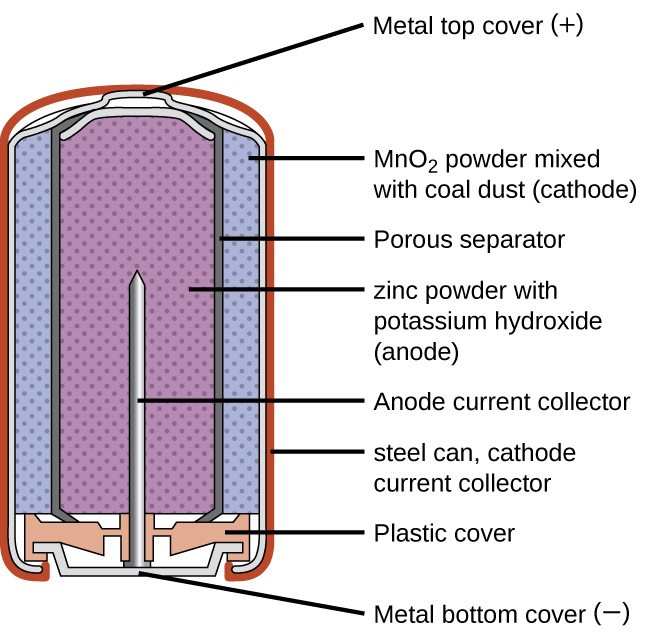
Alkaline batteries (Figure 18.5b) were developed in the 1950s partly to address some of the performance issues with zinc–carbon dry cells. They are manufactured to be exact replacements for zinc-carbon dry cells. As their name suggests, these types of batteries use alkaline electrolytes, often potassium hydroxide. The reactions are
[latex]{\scriptsize \begin{array}{lr @{{}\longrightarrow{}} ll} \text{anode:} & \text{Zn}(s)\;+\;2\text{ OH}^{-}(aq) & \longrightarrow \text{ZnO}(s)\;+\;\text{H}_2\text{O}(l)\;+\;2\text{ e}^{-} & E_{\text{anode}}^{\circ} = -1.28\;\text{V} \\[0.5em] \text{cathode:} & 2\text{ MnO}_2(s)\;+\;\text{H}_2\text{O}(l)\;+\;2\text{ e}^{-} & \longrightarrow \text{Mn}_2\text{O}_3(s)\;+\;2\text{ OH}^{-}(aq) & E_{\text{cathode}}^{\circ} = +0.15\;\text{V} \\[0.5em] \hline \\[-0.25em] \text{overall:} & \text{Zn}(s)\;+\;2\text{ MnO}_2(s) & \longrightarrow \text{ZnO}(s)\;+\;\text{Mn}_2\text{O}_3(s) & E_{\text{cell}}^{\circ} = +1.43\;\text{V} \end{array} \scriptsize}[/latex]
An alkaline battery can deliver about three to five times the energy of a zinc-carbon dry cell of similar size. Alkaline batteries are prone to leaking potassium hydroxide, so these should also be removed from devices for long-term storage. While some alkaline batteries are rechargeable, most are not. Attempts to recharge an alkaline battery that is not rechargeable often leads to rupture of the battery and leakage of the potassium hydroxide electrolyte.
Secondary Batteries
Secondary batteries are rechargeable. These are the types of batteries found in devices such as smartphones, electronic tablets, and automobiles.
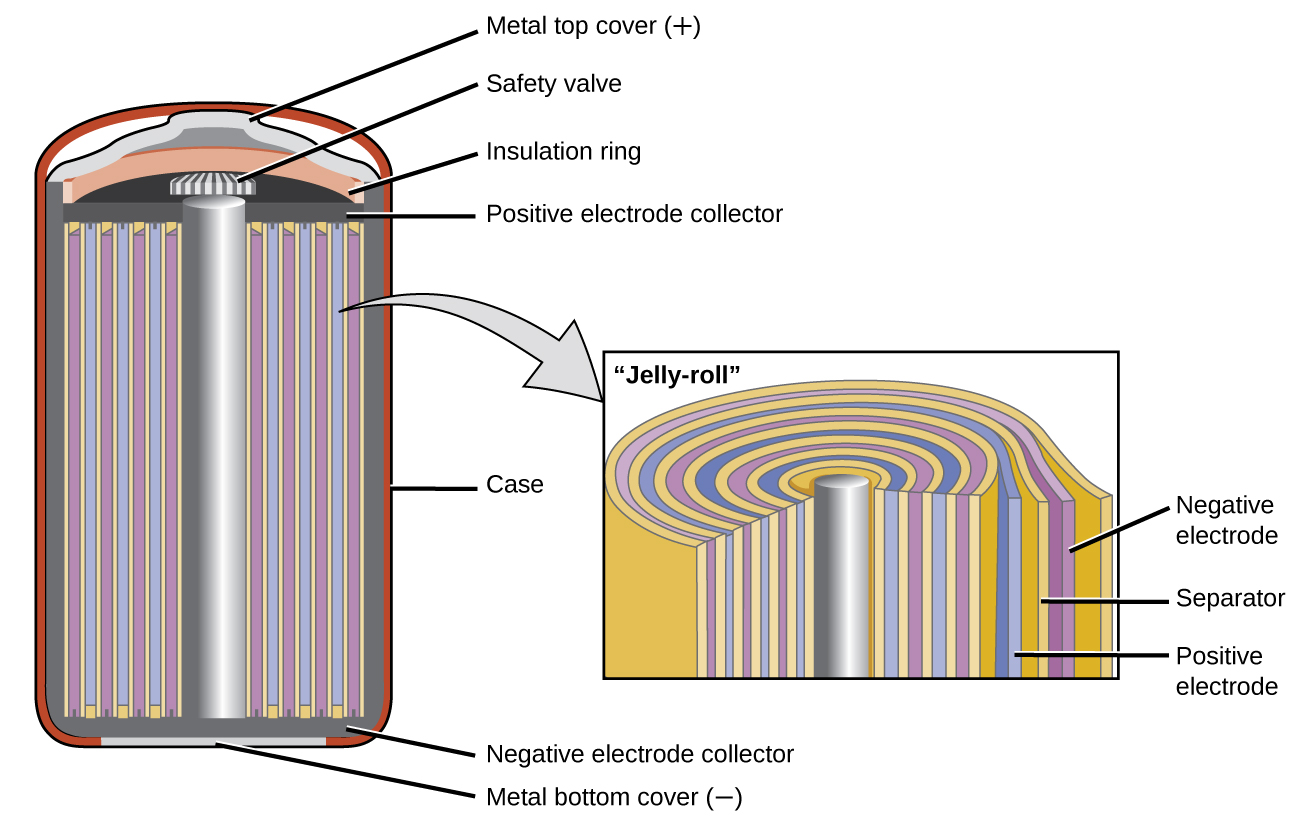
Nickel-cadmium, or NiCd, batteries (Figure 18.5c) consist of a nickel-plated cathode, cadmium-plated anode, and a potassium hydroxide electrode. The positive and negative plates, which are prevented from shorting by the separator, are rolled together and put into the case. This is a “jelly-roll” design and allows the NiCd cell to deliver much more current than a similar-sized alkaline battery. The reactions are
[latex]\begin{array}{lr @{{}\longrightarrow{}} l} \text{anode:} & \text{Cd}(s)\;+\;2\text{ OH}^{-}(aq) & \longrightarrow \text{Cd(OH)}_2(s)\;+\;2\text{ e}^{-} \\[0.5em] \text{cathode:} & \text{NiO}_2(s)\;+\;2\text{ H}_2\text{O}(l)\;+\;2\text{ e}^{-} & \longrightarrow \text{Ni(OH)}_2(s)\;+\;2\text{ OH}^{-}(aq) \\[0.5em] \hline \\[-0.25em] \text{overall:} & \text{Cd}(s)\;+\;\text{NiO}_2(s)\;+\;2\text{ H}_2\text{O}(l) & \longrightarrow \text{Cd(OH)}_2(s)\;+\;\text{Ni(OH)}_2(s) \end{array}[/latex]
The voltage is about 1.2 V to 1.25 V as the battery discharges. When properly treated, a NiCd battery can be recharged about 1000 times. Cadmium is a toxic heavy metal so NiCd batteries should never be opened or put into the regular trash.
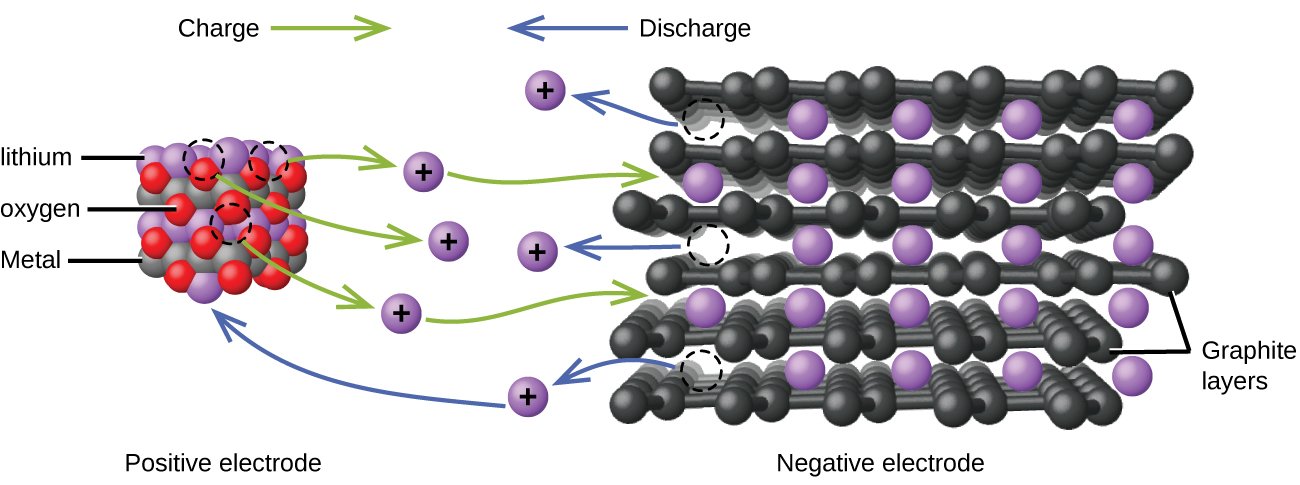
Lithium ion batteries (Figure 18.5d) are among the most popular rechargeable batteries and are used in many portable electronic devices. The reactions are
[latex]\begin{array}{lr @{{}\leftrightharpoons{}} l} \text{anode:} & \text{LiCoO}_2 & \leftrightharpoons \text{Li}_{x\;-\;1}\text{CoO}_2\;+\;x\;\text{Li}^{+}\;+\;x\;\text{e}^{-} \\[0.5em] \text{cathode:} & x\;\text{Li}^{+}\;+\;x\;\text{e}^{-}\;+\;x\;\text{C}_6 & \leftrightharpoons x\;\text{LiC}_6 \\[0.5em] \hline \\[-0.25em] \text{overall:} & \text{LiCoO}_2\;+\;x\;\text{C}_6 & \leftrightharpoons \text{Li}_{x\;-\;1}\text{CoO}_2\;+\;x\;\text{LiC}_6 \end{array}[/latex]
With the coefficients representing moles, x is no more than about 0.5 moles. The battery voltage is about 3.7 V. Lithium batteries are popular because they can provide a large amount of current, are lighter than comparable batteries of other types, produce a nearly constant voltage as they discharge, and only slowly lose their charge when stored.
The lead acid battery (Figure 18.5e) is the type of secondary battery used in your automobile. It is inexpensive and capable of producing the high current required by automobile starter motors. The reactions for a lead acid battery are
Each cell produces 2 V, so six cells are connected in series to produce a 12-V car battery. Lead acid batteries are heavy and contain a caustic liquid electrolyte, but are often still the battery of choice because of their high current density. Since these batteries contain a significant amount of lead, they must always be disposed of properly.
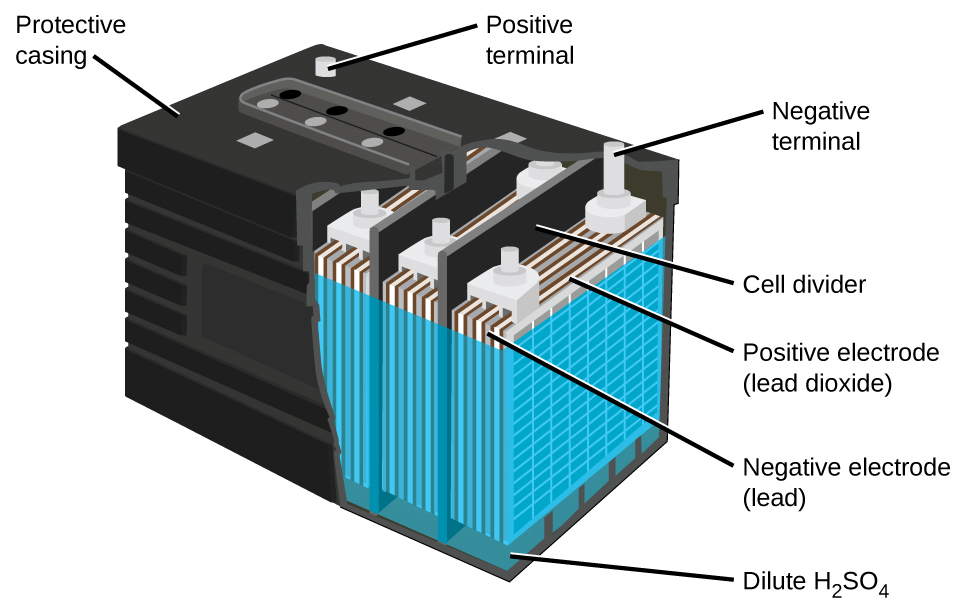
Watch How a lead-acid battery works (4:55 min)
Video source: Engineerguy. (2012, July 3). How a lead-acid battery works [Video]. YouTube.
Fuel Cells
A fuel cell is a device that converts chemical energy into electrical energy. Fuel cells are similar to batteries but require a continuous source of fuel, often hydrogen. They will continue to produce electricity as long as fuel is available. Hydrogen fuel cells have been used to supply power for satellites, space capsules, automobiles, boats, and submarines (Figure 18.5f).
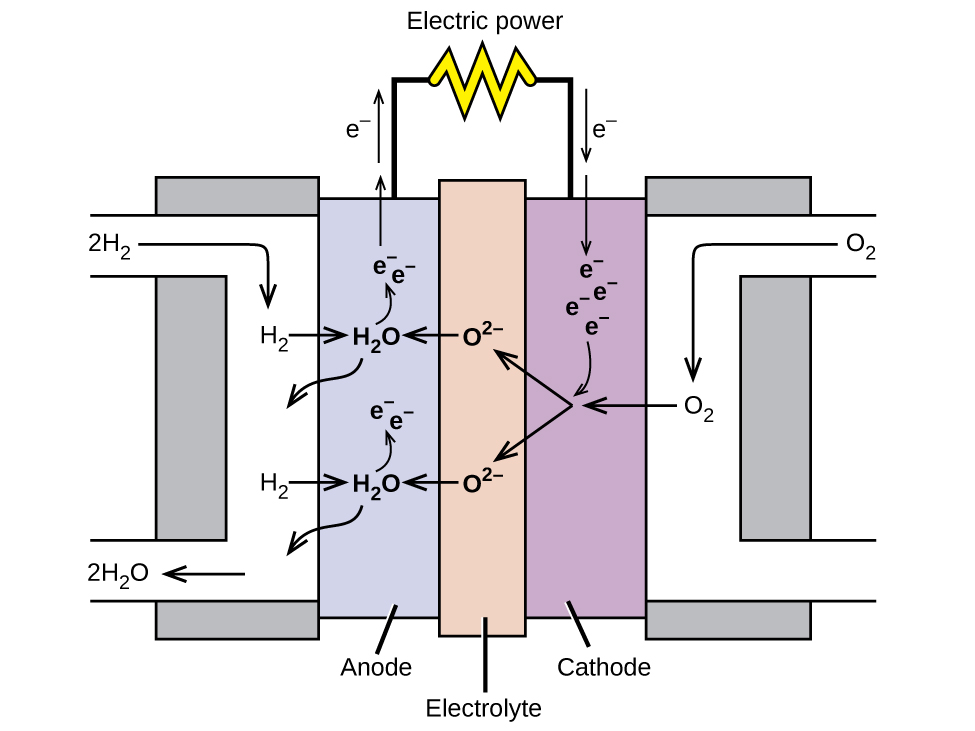
In a hydrogen fuel cell, the reactions are
[latex]\begin{array}{lr @{{}\longrightarrow{}} l} \text{anode:} & 2\text{ H}_2\;+\;2\text{ O}^{2-} & \longrightarrow 2\text{ H}_2\text{O}\;+\;4\text{ e}^{-} \\[0.5em] \text{cathode:} & \text{O}_2\;+\;4\text{ e}^{-} & \longrightarrow 2\text{ O}^{2-} \\[0.5em] \hline \\[-0.25em] \text{overall:} & 2\text{ H}_2\;+\;\text{O}_2 & \longrightarrow 2\text{ H}_2\text{O} \end{array}[/latex]
The voltage is about 0.9 V. The efficiency of fuel cells is typically about 40% to 60%, which is higher than the typical internal combustion engine (25% to 35%) and, in the case of the hydrogen fuel cell, produces only water as exhaust. Currently, fuel cells are rather expensive and contain features that cause them to fail after a relatively short time.
Watch Hydrogen Fuel Cell (1:17 min)
Video source: Jeff Bennett. (2009, October 28). Hydrogen fuel cell [Video]. YouTube.
Attribution & References
Except where otherwise noted, this page is adapted by David Wegman from "17.5 Batteries and Fuel Cells" In General Chemistry 1 & 2 by Rice University, a derivative of Chemistry (Open Stax) by Paul Flowers, Klaus Theopold, Richard Langley & William R. Robinson and is licensed under CC BY 4.0. Access for free at Chemistry (OpenStax).
galvanic cell or series of cells that produces a current; in theory, any galvanic cell
single-use nonrechargeable battery
primary battery, also called a zinc-carbon battery; can be used in any orientation because it uses a paste as the electrolyte; tends to leak electrolyte when stored
primary battery that uses an alkaline (often potassium hydroxide) electrolyte; designed to be an exact replacement for the dry cell, but with more energy storage and less electrolyte leakage than typical dry cell
battery that can be recharged
(NiCd battery) secondary battery that uses cadmium, which is a toxic heavy metal; heavier than lithium ion batteries, but with similar performance characteristics
very popular secondary battery; uses lithium ions to conduct current and is light, rechargeable, and produces a nearly constant potential as it discharges
secondary battery that consists of multiple cells; the lead acid battery found in automobiles has six cells and a voltage of 12 V
devices that produce an electrical current as long as fuel and oxidizer are continuously added; more efficient than internal combustion engines

