Appendix J: Ionization Constants of Weak Bases
Ionization Constants of Weak Bases
| Base | Lewis Structure | Kb at 25 °C |
|---|---|---|
| ammonia | 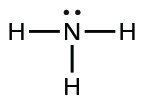 |
1.8 × 10−5 |
| dimethylamine | 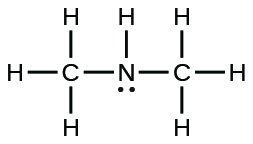 |
5.9 × 10−4 |
| methylamine | 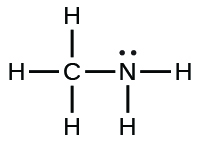 |
4.4 × 10−4 |
| phenylamine (aniline) | 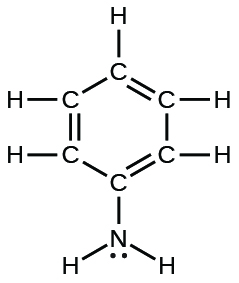 |
4.3 × 10−10 |
| trimethylamine | 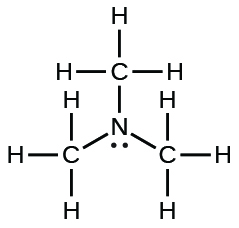 |
6.3 × 10−5 |
Attribution & References
Except where otherwise noted, this page is adapted from “Appendix I: Ionization Constants of Weak Bases” In General Chemistry 1 & 2 by Rice University, a derivative of Chemistry (Open Stax) by Paul Flowers, Klaus Theopold, Richard Langley & William R. Robinson and is licensed under CC BY 4.0. Access for free at Chemistry (OpenStax)

