2.7 – Diffusion and Effusion
You’re hanging out with your friends waiting for a pizza to arrive. The doorbell rings, and one of your friends gets up to open the door and receive the piping hot goodness. Just several seconds after and perhaps even before the door opens, you can already smell its amazing aroma. The quick speed at which the aroma propagates from the door to your nose is due to the nature of gases – gaseous molecules can quickly spread throughout a room. In this section, we discuss the process of diffusion and effusion of gases as an application of the KMT.
Diffusion
Although gaseous molecules travel at tremendous speeds (hundreds of meters per second), they collide with other gaseous molecules and travel in many different directions before reaching the desired target. At room temperature, a gaseous molecule will experience billions of collisions per second. The mean free path is the average distance a molecule travels between collisions. The mean free path increases with decreasing pressure; in general, the mean free path for a gaseous molecule will be hundreds of times the diameter of the molecule.
In general, we know that when a sample of gas is introduced to one part of a closed container, its molecules very quickly disperse throughout the container; this process by which molecules disperse in space in response to differences in concentration is called diffusion (shown in Figure 2.7.1). The gaseous atoms or molecules are, of course, unaware of any concentration gradient, they simply move randomly—regions of higher concentration have more particles than regions of lower concentrations, and so a net movement of species from high to low concentration areas takes place. In a closed environment, diffusion will ultimately result in equal concentrations of gas throughout, as depicted in Figure 2.7.1. The gaseous atoms and molecules continue to move, but since their concentrations are the same in both bulbs, the rates of transfer between the bulbs are equal (no net transfer of molecules occurs).
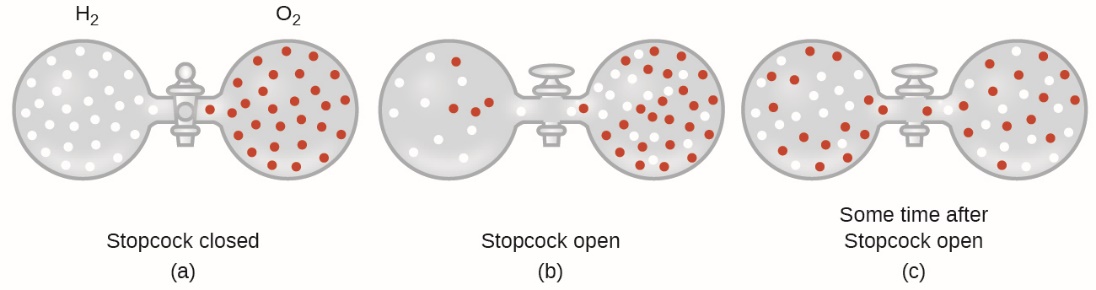
Figure 2.7.1. (a) Two gases, H2 and O2, are initially separated. (b) When the stopcock is opened, they mix together. The lighter gas, H2, passes through the opening faster than O2, so just after the stopcock is opened, more H2 molecules move to the O2 side than O2 molecules move to the H2 side. (c) After a short time, both the slower-moving O2 molecules and the faster-moving H2 molecules have distributed themselves evenly on both sides of the vessel.
We are often interested in the rate of diffusion, the amount of gas passing through some area per unit time:
![]()
Equation 2.7.1 Rate of Diffusion
The diffusion rate depends on several factors: the concentration gradient (the increase or decrease in concentration from one point to another); the amount of surface area available for diffusion; and the distance the gas particles must travel. Note also that the time required for diffusion to occur is inversely proportional to the rate of diffusion, as shown in the rate of diffusion equation.
Effusion
A process involving movement of gaseous species similar to diffusion is effusion, the escape of gas molecules through a tiny hole such as a pinhole in a balloon into a vacuum (Figure 2.7.2). Although diffusion and effusion rates both depend on the molar mass of the gas involved, their rates are not equal; however, the ratios of their rates are identical.
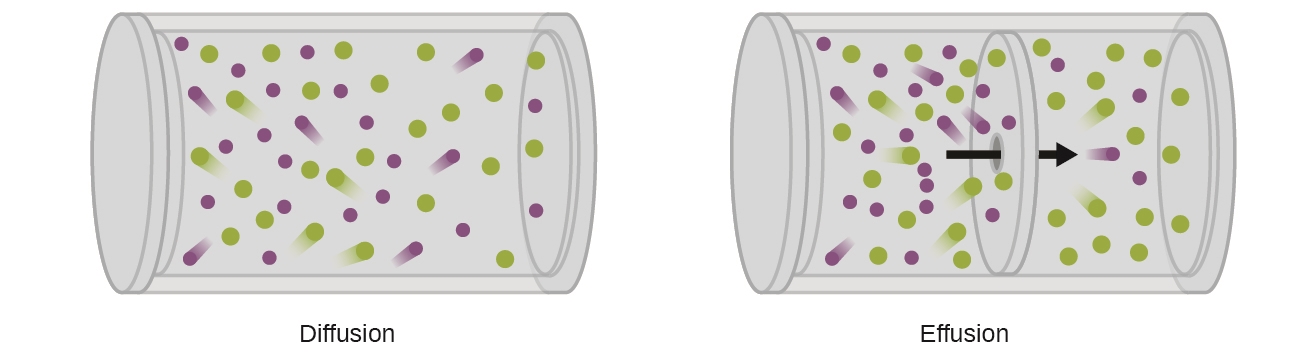
Figure 2.7.2. Diffusion occurs when gas molecules disperse throughout a container. Effusion occurs when a gas passes through an opening that is smaller than the mean free path of the particles, that is, the average distance traveled between collisions. Effectively, this means that only one particle passes through at a time.
If two gases of differing particle masses are placed into separate enclosed chambers, each with a wall containing a single pinhole, both gases will gradually effuse through the small opening in the wall. However, the lighter gas passes through the small opening more rapidly (at a higher rate) than the heavier one (Figure 2.7.3). In 1832, Thomas Graham studied the rates of effusion of different gases and formulated Graham’s law of effusion: The rate of effusion of a gas is inversely proportional to the square root of the mass of its particles:
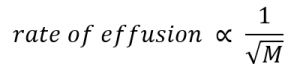
Equation 2.7.2 Rate of effusion
This means that if two gases A and B are at the same temperature and pressure, the ratio of their effusion rates is inversely proportional to the ratio of the square roots of the masses of their particles:
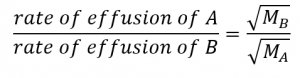
Note that, when Graham’s Law is used in this form, you can use molar masses expressed in either kg/mol or g/mol, as the units will cancel one another. Similarly, other parameters which are directly proportional to the gas’s mass, such as density, can also be used.
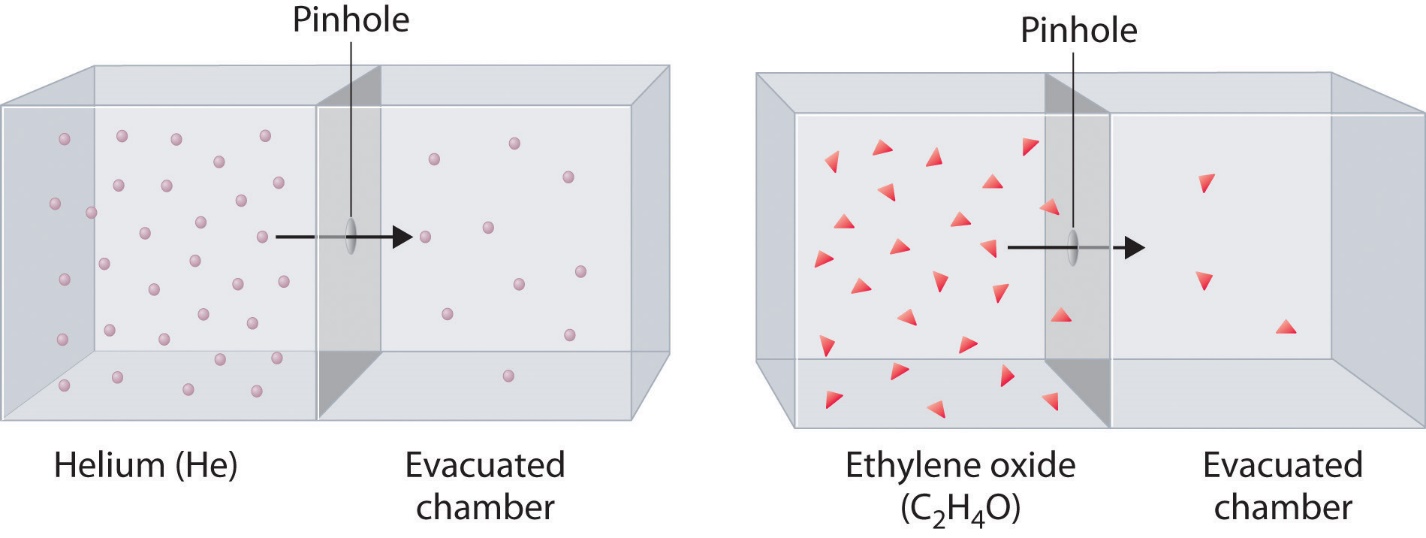
Figure 2.7.3. The lighter He atoms (M = 4.00 g/mol) effuse through the small hole more rapidly than the heavier ethylene oxide (C2H4O) molecules (M = 44.0 g/mol), as predicted by Graham’s law.
The Kinetic-Molecular Theory Explains the Behavior of Gases, Part II
According to Graham’s law (discussed next in this section), the molecules of a gas are in rapid motion and the molecules themselves are small. The average distance between the molecules of a gas is large compared to the size of the molecules. As a consequence, gas molecules can move past each other easily and diffuse at relatively fast rates. The rate of effusion of a gas depends directly on the (root mean average) speed of its molecules:
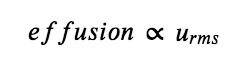
Using this relation, and the equation relating molecular speed to mass, Graham’s law may be easily derived as shown here, for 2 gases A and B:
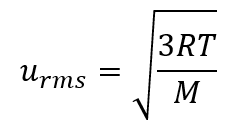
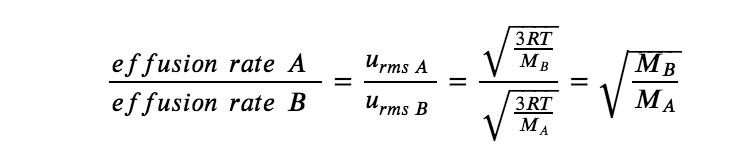
The ratio of the rates of effusion is thus derived to be inversely proportional to the ratio of the square roots of their masses. This is the same relation observed experimentally and expressed as Graham’s law. Check out this video to better visualize rates of effusion.
Example 2.7.1 – Applying Graham’s Law to Rates of Effusion
Calculate the ratio of the rate of effusion of hydrogen to the rate of effusion of oxygen, using a) molar masses; and b) gas densities.
Solution
Using Graham’s Law and applying molar masses:
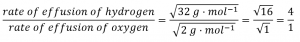
The densities of oxygen and hydrogen at STP are 1.43 g/L and 0.0899 g/L,
respectively. Recalculating Graham’s Law with densities gives the identical
numerical result:
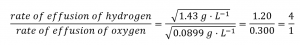
Hydrogen effuses four times as rapidly as oxygen.
Check Your Learning 2.7.1 – Applying Graham’s Law to Rates of Effusion
At a particular pressure and temperature, nitrogen gas effuses at the rate of 79 mL/s. Using the same apparatus at the same temperature and pressure, at what rate will sulfur dioxide effuse?
Answer:
52 mL/s
Here’s another example, making the point about how determining times differs from determining rates.
Example 2.7.2 – Effusion Time Calculations
It takes 243 s for 4.46 × 10−5 mol Xe to effuse through a tiny hole. Under the same conditions, how long will it take 4.46 × 10−5 mol Ne to effuse?
Solution
It is important to resist the temptation to use the amounts of time directly, and to remember instead how rate relates to time as well as how it relates to mass. Recall the definition of rate of effusion:
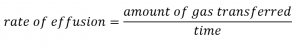
and combine it with Graham’s law:
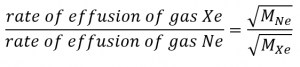
To get:
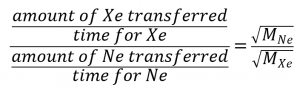
Note that amount of A = amount of B, which simplifies the equation:
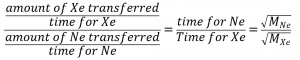
Finally, substitute values and solve for the desired quantity : 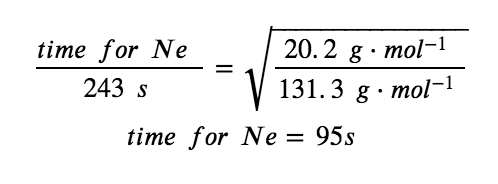
Note that this answer is reasonable: Since Ne is lighter than Xe, the effusion rate for Ne will be larger than that for Xe, which means the time of effusion for Ne will be smaller than that for Xe.
Check Your Learning 2.7.2 – Effusion Time Calculations
A party balloon filled with helium deflates to 2/3 of its original volume in 8.0 hours. How long will it take an identical balloon filled with the same number of moles of air (Mm = 28.2 g/mol) to deflate to 1/2 of its original volume?
Answer:
32 h
Finally, here is one more example showing how to calculate molar mass from effusion rate data.
Example 2.7.3 – Determining Molar Mass Using Graham’s Law
An unknown gas effuses about 1.21 times more rapidly than CO2. What is the molar mass of the unknown gas? Of the following 5 gases, which best fits the identity of the unknown gas??
Ne NO CH4 C3H8 SO2
Solution
From Graham’s law, we have:
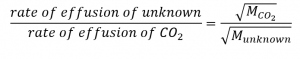
Plug in known data:
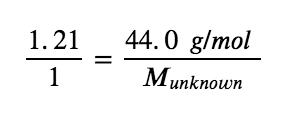
Solve:
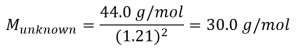
The molar masses of the five gaseous compounds listed are:
Ne CH4 NO C3H8 SO2
20.18 g/mol 16.04 g/mol 30.01 g/mol 44.09 g/mol 64.06 g/mol
Of the five compounds, NO, also known as nitric oxide, has a molar mass of 30.01 g/mol. Hence, from the choices provided, the gas is NO.
Check Your Learning 2.7.3 – Determining Molar Mass Using Graham’s Law
Hydrogen gas effuses through a porous container 8.97-times faster than an unknown gas. Estimate the molar mass of the unknown gas.
Answer
163 g/mol
|
In Case You’re Interested… Use of Diffusion for Nuclear Energy Applications: Uranium Enrichment |
|
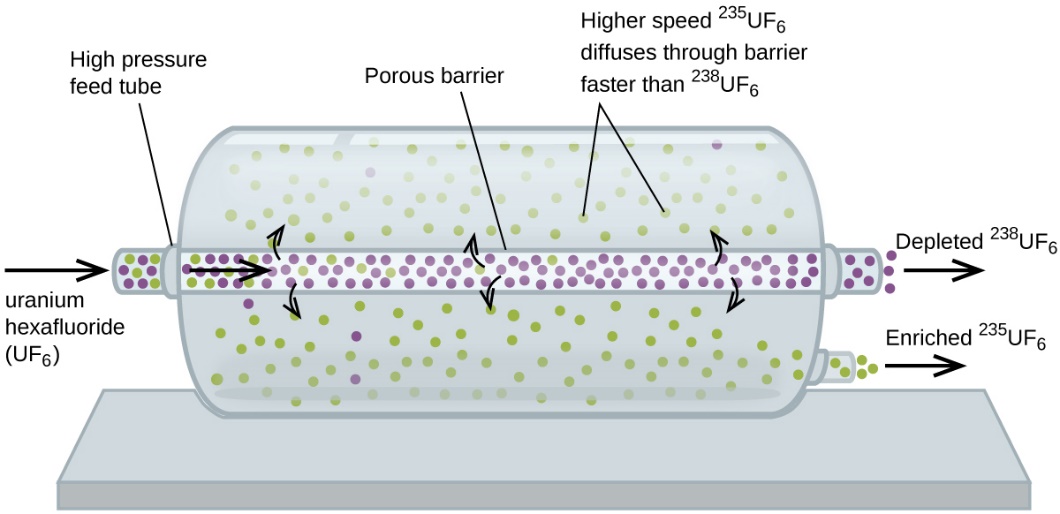
Gaseous diffusion has been used to produce enriched uranium for use in nuclear power plants and weapons. Naturally occurring uranium contains only 0.72% of 235U, the kind of uranium that is “fissile,” that is, capable of sustaining a nuclear fission chain reaction. Nuclear reactors require fuel that is 2–5% 235U, and nuclear bombs need even higher concentrations. One way to enrich uranium to the desired levels is to take advantage of Graham’s law. In a gaseous diffusion enrichment plant, uranium hexafluoride (UF6, the only uranium compound that is volatile enough to work) is slowly pumped through large cylindrical vessels called diffusers, which contain porous barriers with microscopic openings. The process is one of diffusion because the other side of the barrier is not evacuated. The 235UF6 molecules have a higher average speed and diffuse through the barrier a little faster than the heavier 238UF6 molecules. The gas that has passed through the barrier is thus slightly enriched in 235UF6 and the residual gas is slightly depleted. The small difference in molecular weights between 235UF6 and 238UF6 results in only about 0.4% enrichment per diffuser (Figure 2.7.4). But by connecting many diffusers in a sequence of stages (called a cascade), the desired level of enrichment can be attained.
Figure 2.7.4. In a diffuser, gaseous UF6 is pumped through a porous barrier, which partially separates 235UF6 from 238UF6 The UF6 must pass through many large diffuser units to achieve sufficient enrichment in 235U. The large-scale separation of gaseous 235UF6 from 238UF6 was first done during World War II, at the atomic energy installation in Oak Ridge, Tennessee, as part of the Manhattan Project (the development of the first atomic bomb). Although the theory is simple, this required surmounting many daunting technical challenges to make it work in practice. The barrier must have tiny, uniform holes (about 10–6 cm in diameter) and be porous enough to produce high flow rates. All materials (the barrier, tubing, surface coatings, lubricants, and gaskets) need to be able to contain, but not react with, the highly reactive and corrosive UF6. Because gaseous diffusion plants require very large amounts of energy (to compress the gas to the high pressures required and drive it through the diffuser cascade, to remove the heat produced during compression, and so on), it is now being replaced by gas centrifuge technology, which requires far less energy. |
Questions
★ Questions
1. A balloon filled with helium gas is found to take 6 hours to deflate to 50% of its original volume. How long will it take (time) for an identical balloon filled with the same volume of hydrogen gas (instead of helium) to decrease its volume by 50% (in hours)?
2. Explain why the numbers of molecules are not identical in the left- and right-hand bulbs shown in the center illustration of Figure 2.7.1.
3. Starting with the definition of rate of effusion and Graham’s finding relating rate and molar mass, show how to derive the Graham’s law equation, relating the relative rates of effusion for two gases to their molecular masses.
4. Heavy water, D2O (molar mass = 20.03 g mol–1), can be separated from ordinary water, H2O (molar mass = 18.01), as a result of the difference in the relative rates of diffusion of the molecules in the gas phase. Calculate the relative rates of diffusion of H2O and D2O.
5. Which of the following gases diffuse more slowly than oxygen? F2, Ne, N2O, C2H2, NO, Cl2, H2S
★★ Questions
6. During the discussion of gaseous diffusion for enriching uranium, it was claimed that 235UF6 diffuses 0.4% faster than 238UF6. Show the calculation that supports this value. The molar mass of 235UF6 = 235.043930 + 6 × 18.998403 = 349.034348 g/mol, and the molar mass of 238UF6 = 238.050788 + 6 × 18.998403 = 352.041206 g/mol.
7. Calculate the relative rate of diffusion of 1H2 (molar mass 2.0 g/mol) compared to that of 2H2 (molar mass 4.0 g/mol) and the relative rate of diffusion of O2 (molar mass 32 g/mol) compared to that of O3 (molar mass 48 g/mol).
8. A gas of unknown identity diffuses at a rate of 83.3 mL/s in a diffusion apparatus in which carbon dioxide diffuses at the rate of 102 mL/s. Calculate the molecular mass (g⋅mol-1) of the unknown gas.
9. When two cotton plugs, one moistened with ammonia and the other with hydrochloric acid, are simultaneously inserted into opposite ends of a glass tube that is 87.0 cm long, a white ring of NH4Cl forms where gaseous NH3 and gaseous HCl first come into contact.
NH3 (g) + HCl (g) ⟶ NH4Cl (s)
At approximately what distance from the ammonia moistened plug does this occur? (Hint: Calculate the rates of diffusion for both NH3 and HCl, and find out how much faster NH3 diffuses than HCl.)
Answers
1. 4.2 hours
2. In the centre figure, it is seen that there is more H2 present on the right side versus the left side. The reason for this is the rate of effusion. H2 has a faster rate of effusion in comparison to O2 as H2 has a smaller molecular mass. This means that H2 will pass through the hole at a faster rate than O2 which creates that unequal particle distribution seen in Figure 2.7.1.
3. Effusion can be defined as the process by which a gas escapes through a pinhole into a vacuum. Graham’s law states that with a mixture of two gases A and B: ![]() . Both A and B are in the same container at the same temperature and therefore will have the same kinetic energy:
. Both A and B are in the same container at the same temperature and therefore will have the same kinetic energy:
![]() Therefore,
Therefore,
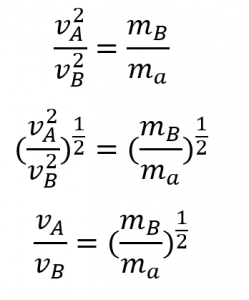
4. ![]()
5. F2, N2O, Cl2, H2S
6.

7. 1.4; 1.2
8. 65.99 g mol-1
9. 51.7 cm
Average distance a molecule travels between collisions
Movement of an atom or molecule from a region of relatively high concentration to one of relatively low concentration (discussed in Chapter 2 with regard to gaseous species, but applicable to species in any phase)
Amount of gas diffusing through a given area over a given time
Transfer of gaseous atoms or molecules from a container to a vacuum through very small openings
Rates of diffusion and effusion of gases are inversely proportional to the square roots of their molecular masses