21. Laboratory Analysis of the Immune Response
21.4 EIAs and ELISAs
Learning Objectives
- Explain the differences and similarities between EIA, FEIA, and ELISA
- Describe the difference and similarities between immunohistochemistry and immunocytochemistry
- Describe the different purposes of direct and indirect ELISA
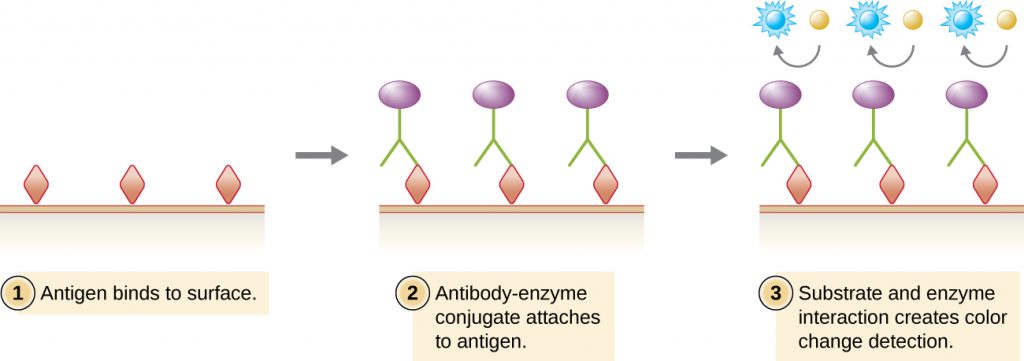
Similar to the western blot, enzyme immunoassays (EIAs) use antibodies to detect the presence of antigens. However, EIAs differ from western blots in that the assays are conducted in microtitre plates or in vivo rather than on an absorbent membrane. There are many different types of EIAs, but they all involve an antibody molecule whose constant region binds an enzyme, leaving the variable region free to bind its specific antigen. The addition of a substrate for the enzyme allows the antigen to be visualized or quantified (Figure 21.22).
In EIAs, the substrate for the enzyme is most often a chromogen, a colourless molecule that is converted into a coloured end product. The most widely used enzymes are alkaline phosphatase and horseradish peroxidase for which appropriate substrates are readily available. In some EIAs, the substrate is a fluorogen, a non-fluorescent molecule that the enzyme converts into a fluorescent form. EIAs that utilize a fluorogen are called fluorescent enzyme immunoassays (FEIAs). Fluorescence can be detected by either a fluorescence microscope or a spectrophotometer.
MICRO CONNECTIONS: The MMR Titre
The MMR vaccine is a combination vaccine that provides protection against measles, mumps, and rubella (German measles). Most people receive the MMR vaccine as children and thus have antibodies against these diseases. However, for various reasons, even vaccinated individuals may become susceptible to these diseases again later in life. For example, some children may receive only one round of the MMR vaccine instead of the recommended two. In addition, the titre of protective antibodies in an individual’s body may begin to decline with age or as the result of some medical conditions.
To determine whether the titre of antibody in an individual’s bloodstream is sufficient to provide protection, an MMR titre test can be performed. The test is a simple immunoassay that can be done quickly with a blood sample. The results of the test will indicate whether the individual still has immunity or needs another dose of the MMR vaccine.
Submitting to an MMR titre is often a pre-employment requirement for healthcare workers, especially those who will frequently be in contact with young children or immunocompromised patients. Were a healthcare worker to become infected with measles, mumps, or rubella, the individual could easily pass these diseases on to susceptible patients, leading to an outbreak. Depending on the results of the MMR titre, healthcare workers might need to be revaccinated prior to beginning work.
Immunostaining
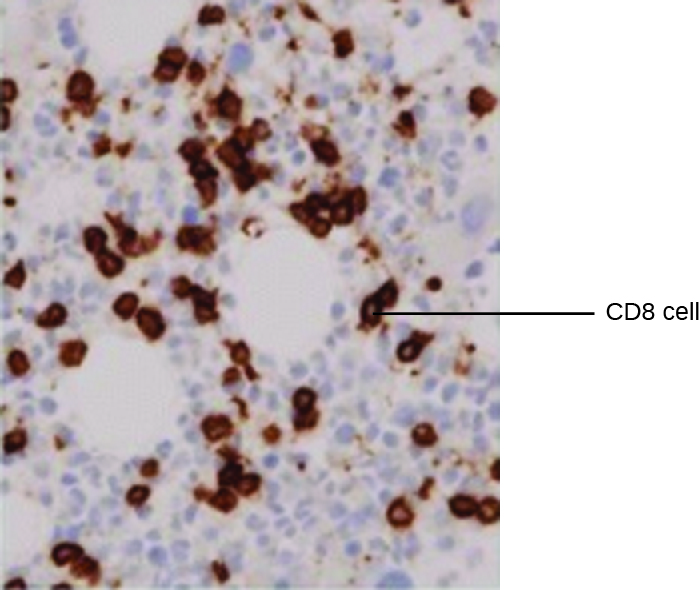
One powerful use of EIA is immunostaining, in which antibody-enzyme conjugates enhance microscopy. Immunohistochemistry (IHC) is used for examining whole tissues. As seen in Figure 21.23, a section of tissue can be stained to visualize the various cell types. In this example, a mAb against CD8 was used to stain CD8 cells in a section of tonsil tissue. It is now possible to count the number of CD8 cells, determine their relative numbers versus the other cell types present, and determine the location of these cells within this tissue. Such data would be useful for studying diseases such as AIDS, in which the normal function of CD8 cells is crucial for slowing disease progression.
Immunocytochemistry (ICC) is another valuable form of immunostaining. While similar to IHC, in ICC, extracellular matrix material is stripped away, and the cell membrane is etched with alcohol to make it permeable to antibodies. This allows antibodies to pass through the cell membrane and bind to specific targets inside the cell. Organelles, cytoskeletal components, and other intracellular structures can be visualized in this way. While some ICC techniques use EIA, the enzyme can be replaced with a fluorescent molecule, making it a fluorescent immunoassay.
- What is the difference between immunohistochemistry and immunocytochemistry?
- What must be true of the product of the enzymatic reaction used in immunohistochemistry?
Enzyme-linked Immunosorbent Assays (ELISAs)
The enzyme-linked immunosorbent assays (ELISAs) are widely used EIAs. In the direct ELISA, antigens are immobilized in the well of a microtitre plate. An antibody that is specific for a particular antigen and is conjugated to an enzyme is added to each well. If the antigen is present, then the antibody will bind. After washing to remove any unbound antibodies, a colourless substrate (chromogen) is added. The presence of the enzyme converts the substrate into a coloured end product (Figure 21.22). While this technique is faster because it only requires the use of one antibody, it has the disadvantage that the signal from a direct ELISA is lower (lower sensitivity).
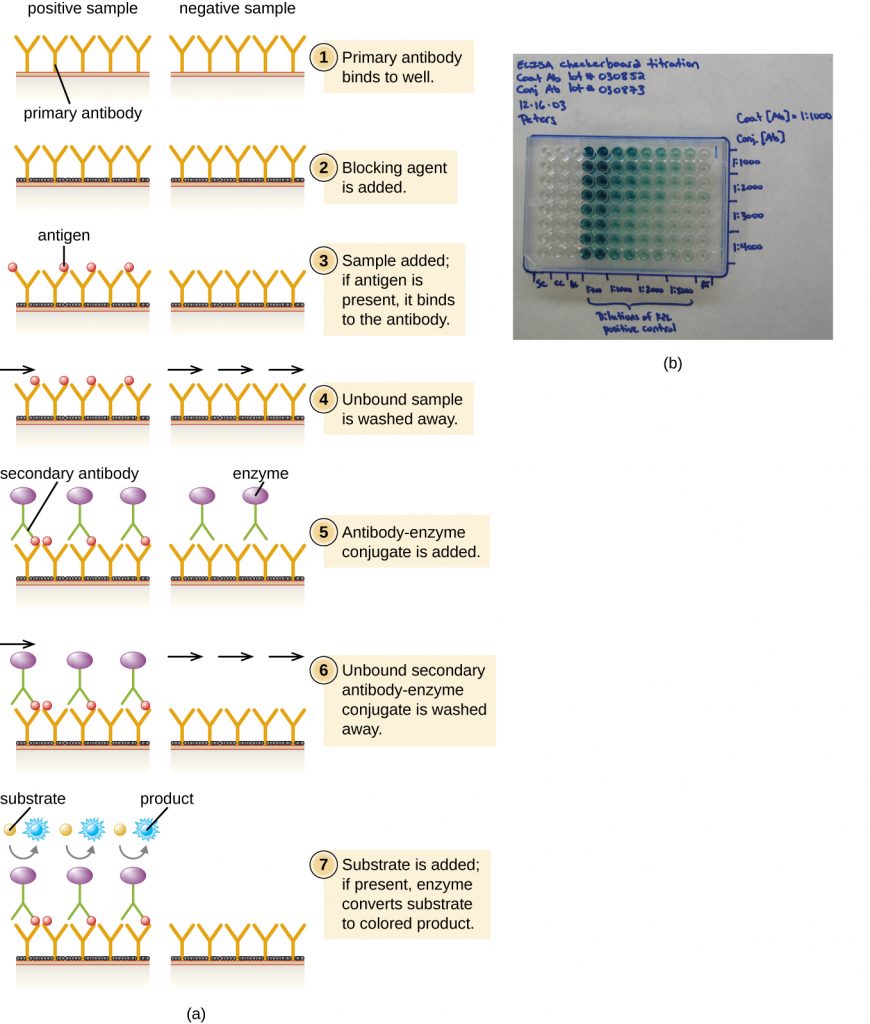
In a sandwich ELISA, the goal is to use antibodies to precisely quantify specific antigen present in a solution, such as antigen from a pathogen, a serum protein, or a hormone from the blood or urine to list just a few examples. The first step of a sandwich ELISA is to add the primary antibody to all the wells of a microtitre plate (Figure 21.24). The antibody sticks to the plastic by hydrophobic interactions. After an appropriate incubation time, any unbound antibody is washed away. Comparable washes are used between each of the subsequent steps to ensure that only specifically bound molecules remain attached to the plate. A blocking protein is then added (e.g., albumin or the milk protein casein) to bind the remaining nonspecific protein-binding sites in the well. Some of the wells will receive known amounts of antigen to allow the construction of a standard curve, and unknown antigen solutions are added to the other wells. The primary antibody captures the antigen and, following a wash, the secondary antibody is added, which is a polyclonal antibody that is conjugated to an enzyme. After a final wash, a colourless substrate (chromogen) is added, and the enzyme converts it into a coloured end product. The colour intensity of the sample caused by the end product is measured with a spectrophotometer. The amount of colour produced (measured as absorbance) is directly proportional to the amount of enzyme, which in turn is directly proportional to the captured antigen. ELISAs are extremely sensitive, allowing antigen to be quantified in the nanogram (10–9 g) per mL range.
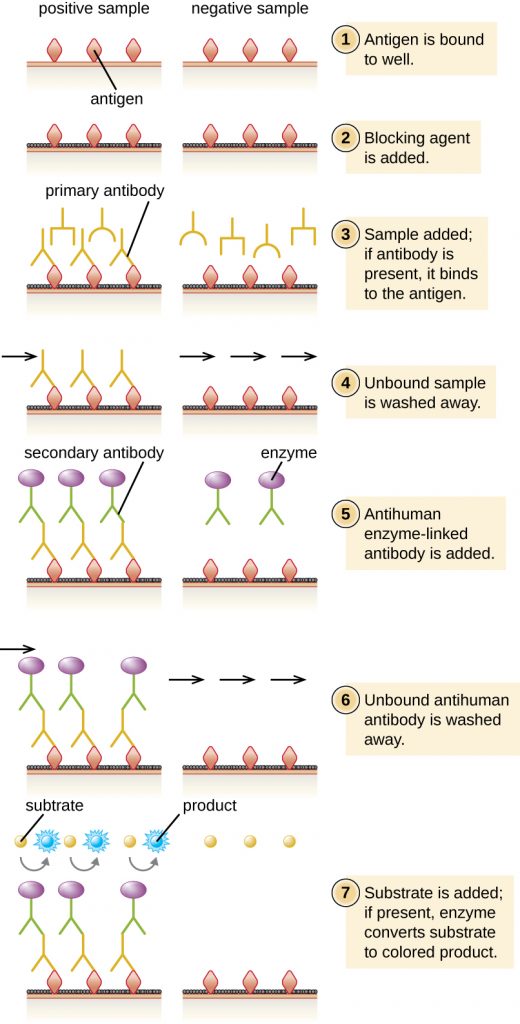
In an indirect ELISA, we quantify antigen-specific antibody rather than antigen. We can use indirect ELISA to detect antibodies against many types of pathogens, including Borrelia burgdorferi (Lyme disease) and HIV. There are three important differences between indirect and direct ELISAs as shown in Figure 21.25. Rather than using antibody to capture antigen, the indirect ELISA starts with attaching known antigen (e.g., peptides from HIV) to the bottom of the microtitre plate wells. After blocking the unbound sites on the plate, patient serum is added; if antibodies are present (primary antibody), they will bind the antigen. After washing away any unbound proteins, the secondary antibody with its conjugated enzyme is directed against the primary antibody (e.g., antihuman immunoglobulin). The secondary antibody allows us to quantify how much antigen-specific antibody is present in the patient’s serum by the intensity of the colour produced from the conjugated enzyme-chromogen reaction.
As with several other tests for antibodies discussed in this chapter, there is always concern about cross-reactivity with antibodies directed against some other antigen, which can lead to false-positive results. Thus, we cannot definitively diagnose an HIV infection (or any other type of infection) based on a single indirect ELISA assay. We must confirm any suspected positive test, which is most often done using either an immunoblot that actually identifies the presence of specific peptides from the pathogen or a test to identify the nucleic acids associated with the pathogen, such as reverse transcriptase PCR (RT-PCR) or a nucleic acid antigen test.
- What is the purpose of the secondary antibody in a direct ELISA?
- What do the direct and indirect ELISAs quantify?
CLINICAL FOCUS: Part 2
Although contacting and testing the 1300 patients for HIV would be time consuming and expensive, administrators hoped to minimize the hospital’s liability by proactively seeking out and treating potential victims of the rogue employee’s crime. Early detection of HIV is important, and prompt treatment can slow the progression of the disease.
There are a variety of screening tests for HIV, but the most widely used is the indirect ELISA. As with other indirect ELISAs, the test works by attaching antigen (in this case, HIV peptides) to a well in a 96-well plate. If the patient is HIV positive, anti-HIV antibodies will bind to the antigen and be identified by the second antibody-enzyme conjugate.
- How accurate is an indirect ELISA test for HIV, and what factors could impact the test’s accuracy?
- Should the hospital use any other tests to confirm the results of the indirect ELISA?
Jump to the previous Clinical Focus box. Jump to the next Clinical Focus box.
Immunofiltration and Immunochromatographic Assays
For some situations, it may be necessary to detect or quantify antigens or antibodies that are present at very low concentration in solution. Immunofiltration techniques have been developed to make this possible. In immunofiltration, a large volume of fluid is passed through a porous membrane into an absorbent pad. An antigen attached to the porous membrane will capture antibody as it passes; alternatively, we can also attach an antibody to the membrane to capture antigen.
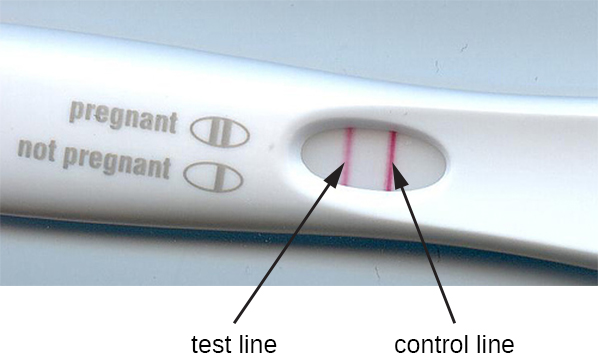
The method of immunofiltration has been adapted in the development of immunochromatographic assays, commonly known as lateral flow tests or strip tests. These tests are quick and easy to perform, making them popular for point-of-care use (i.e., in the doctor’s office) or in-home use. One example is the TORCH test that allows doctors to screen pregnant women or newborns for infection by an array of viruses and other pathogens (Toxoplasma, other viruses, rubella, cytomegalovirus, herpes simplex). In-home pregnancy tests are another widely used example of a lateral flow test (Figure 21.26). Immunofiltration tests are also popular in developing countries, because they are inexpensive and do not require constant refrigeration of the dried reagents. However, the technology is also built into some sophisticated laboratory equipment.
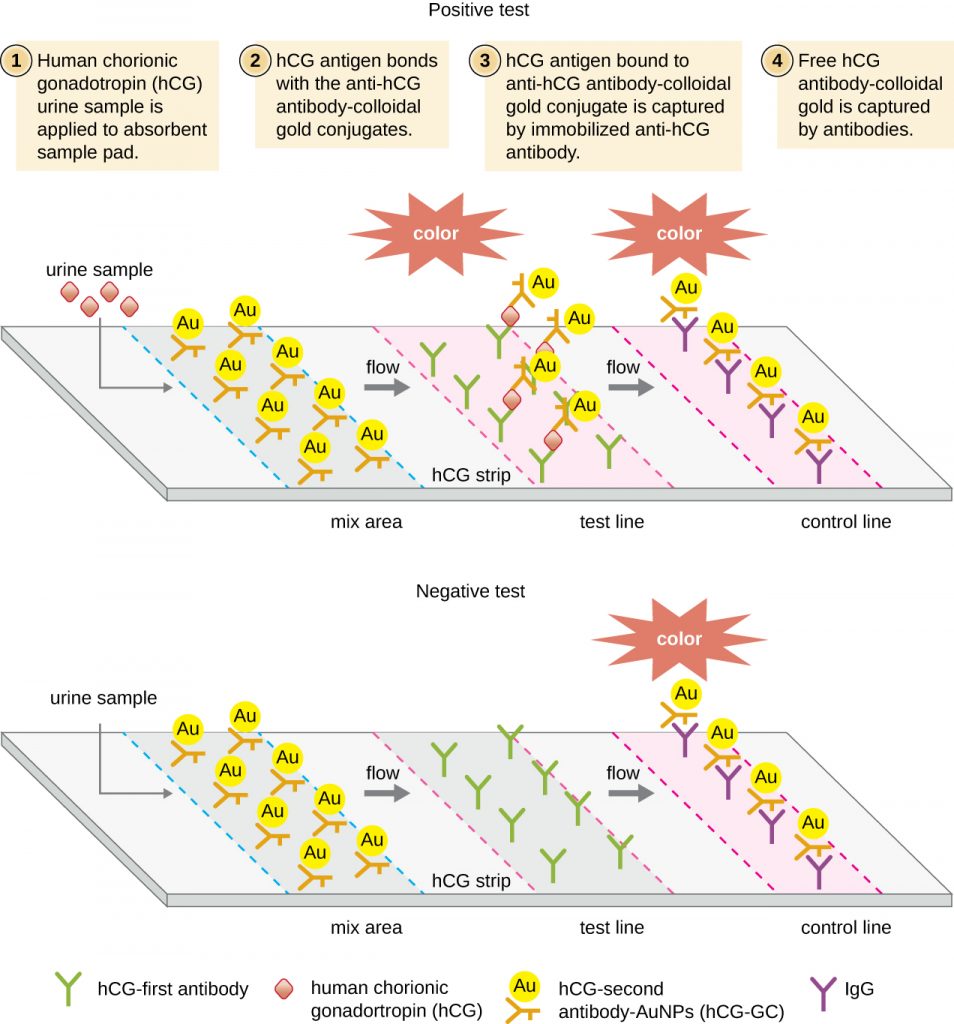
In lateral flow tests (Figure 21.27), fluids such as urine are applied to an absorbent pad on the test strip. The fluid flows by capillary action and moves through a stripe of beads with antibodies attached to their surfaces. The fluid in the sample actually hydrates the reagents, which are present in a dried state in the stripe. Antibody-coated beads made of latex or tiny gold particles will bind antigens in the test fluid. The antibody-antigen complexes then flow over a second stripe that has immobilized antibody against the antigen; this stripe will retain the beads that have bound antigen. A third control stripe binds any beads. A red colour (from gold particles) or blue (from latex beads) developing at the test line indicates a positive test. If the colour only develops at the control line, the test is negative.
Like ELISA techniques, lateral flow tests take advantage of antibody sandwiches, providing sensitivity and specificity. While not as quantitative as ELISA, these tests have the advantage of being fast, inexpensive, and not dependent on special equipment. Thus, they can be performed anywhere by anyone. There are some concerns about putting such powerful diagnostic tests into the hands of people who may not understand the tests’ limitations, such as the possibility of false-positive results. While home pregnancy tests have become widely accepted, at-home antibody-detection tests for diseases like HIV have raised some concerns in the medical community. Some have questioned whether self-administration of such tests should be allowed in the absence of medical personnel who can explain the test results and order appropriate confirmatory tests. However, with growing numbers of lateral flow tests becoming available, and the rapid development of lab-on-a-chip technology (Figure 21.1), home medical tests are likely to become even more commonplace in the future.
- What physical process does the lateral flow method require to function?
- Explain the purpose of the third strip in a lateral flow assay.
Table 21.4 compares some of the key mechanisms and examples of some of the EIAs discussed in this section as well as immunoblots, which were discussed in Sec. 21.2 – Detecting Antigen-Antibody Complexes.
Table 21.4. Immunoblots and Enzyme Immunoassays
| Immunoblots & Enzyme Immunoassays | |||
|---|---|---|---|
| Type of Assay | Mechanism | Specific Procedures | Examples |
| Immunoblots | Uses enzyme-antibody conjugates to identify specific proteins that have been transferred to an absorbent membrane | Western blot: Detects the presence of a particular protein | Detecting the presence of HIV peptides (or peptides from other infectious agents) in patient sera |
| Immunostaining | Uses enzyme-antibody conjugates to stain specific molecules on or in cells | Immunohistochemistry: Used to stain specific cells in a tissue | Stain for presence of CD8 cells in host tissue |
| Enzyme-linked immunosorbent assay (ELISA) | Uses enzyme-antibody conjugates to quantify target molecules | Direct ELISA: Uses a single antibody to detect the presence of an antigen | Detection of HIV antigen p24 up to one month after being infected |
| Indirect ELISA: Measures the amount of antibody produced against an antigen | Detection of HIV antibodies in serum | ||
| Immunochromatographic (lateral flow) assays | Techniques use the capture of flowing, colour-labeled antigen-antibody complexes by fixed antibody for disease diagnosis | Sandwich ELISA: Measures the amount of antigen bound by the antibody | Detection of antibodies for various pathogens in patient sera (e.g., rapid strep, malaria dipstick) |
| Pregnancy test detecting human chorionic gonadotrophin in urine | |||
CLINICAL FOCUS: Part 3
Although the indirect ELISA for HIV is a sensitive assay, there are several complicating considerations. First, if an infected person is tested too soon after becoming infected, the test can yield false-negative results. The seroconversion window is generally about three weeks, but in some cases, it can be more than two months.
In addition to false negatives, false positives can also occur, usually due to previous infections with other viruses that induce cross-reacting antibodies. The false-positive rate depends on the particular brand of test used, but 0.5% is not unusual.[1] Because of the possibility of a false positive, all positive tests are followed up with a confirmatory test. This confirmatory test is often an immunoblot (western blot) in which HIV peptides from the patient’s blood are identified using an HIV-specific mAb-enzyme conjugate. A positive western blot would confirm an HIV infection and a negative blot would confirm the absence of HIV despite the positive ELISA.
Unfortunately, western blots for HIV antigens often yield indeterminate results, in which case, they neither confirm nor invalidate the results of the indirect ELISA. In fact, the rate of indeterminates can be 10–49% (which is why, combined with their cost, western blots are not used for screening). Similar to the indirect ELISA, an indeterminate western blot can occur because of cross-reactivity or previous viral infections, vaccinations, or autoimmune diseases.
- Of the 1300 patients being tested, how many false-positive ELISA tests would be expected?
- Of the false positives, how many indeterminate western blots could be expected?
- How would the hospital address any cases in which a patient’s western blot was indeterminate?
Jump to the previous Clinical Focus box. Jump to the next Clinical Focus box.
Key Takeaways
- Enzyme immunoassays (EIA) are used to visualize and quantify antigens. They use an antibody conjugated to an enzyme to bind the antigen, and the enzyme converts a substrate into an observable end product. The substrate may be either a chromogen or a fluorogen.
- Immunostaining is an EIA technique for visualizing cells in a tissue (immunohistochemistry) or examining intracellular structures (immunocytochemistry).
- Direct ELISA is used to quantify an antigen in solution. The primary antibody captures the antigen, and the secondary antibody delivers an enzyme. Production of end product from the chromogenic substrate is directly proportional to the amount of captured antigen.
- Indirect ELISA is used to detect antibodies in patient serum by attaching antigen to the well of a microtitre plate, allowing the patient (primary) antibody to bind the antigen and an enzyme-conjugated secondary antibody to detect the primary antibody.
- Immunofiltration and immunochromatographic assays are used in lateral flow tests, which can be used to diagnose pregnancy and various diseases by detecting colour-labeled antigen-antibody complexes in urine or other fluid samples
Multiple Choice
Fill in the Blank
Short Answer
- Why is it important in a sandwich ELISA that the antigen has multiple epitopes? And why might it be advantageous to use polyclonal antisera rather than mAb in this assay?
- The pregnancy test strip detects the presence of human chorionic gonadotrophin in urine. This hormone is initially produced by the fetus and later by the placenta. Why is the test strip preferred for this test rather than using either a direct or indirect ELISA with their more quantifiable results?
Critical Thinking
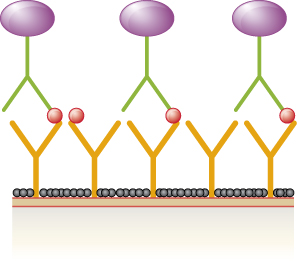
- Label the primary and secondary antibodies, and discuss why the production of end product will be proportional to the amount of antigen.

Media Attributions
- OSC_Microbio_20_04_EIA
- OSC_Microbio_20_04_IHC
- OSC_Microbio_20_04_ELISA
- OSC_Microbio_20_04_Indirect
- OSC_Microbio_20_04_Torch
- OSC_Microbio_20_04_Lateral
- OSC_Microbio_20_04_ArtConnect_img
- Thomas, Justin G., Victor Jaffe, Judith Shaffer, and Jose Abreu, “HIV Testing: US Recommendations 2014,” Osteopathic Family Physician 6, no. 6 (2014). ↵

