The PharmGKB website has excellent resources for examining the pharmacokinetic and pharmacodynamic pathways of various drugs and their pharmacogenomic associations. Below is an example of two commonly prescribed medications, Amitriptyline and Nortriptyline. Search the PharmGKB Pathways for other drugs of interest. For example, returning to our aminoglycoside ototoxicity example from an earlier unit, review the pathway and pharmacogenomic implications.
9.4 Personalized Drug Therapy
Clinical Implications of Pharmacogenomics
What is CPIC®?
The Clinical Pharmacogenetics Implementation Consortium (CPIC®) is an international consortium of individual volunteers and a small dedicated staff interested in facilitating pharmacogenetic test use for patient care.
One barrier to implementing pharmacogenetic testing in the clinic is translating genetic laboratory test results into actionable prescribing decisions for affected drugs.
CPIC® aims to address this barrier to the clinical implementation of pharmacogenetic tests by creating, curating, and posting freely available, peer-reviewed, evidence-based, updatable, and detailed gene/drug clinical practice guidelines (visit their website to view all CPIC® publications). CPIC® guidelines follow standardized formats, include systematic grading of evidence and clinical recommendations, use standardized terminology, are peer-reviewed, and are published in a leading journal (in partnership with Clinical Pharmacology and Therapeutics) with simultaneous posting to cpicpgx.org, where they are regularly updated.
CPIC® started as a shared project between PharmGKB and the Pharmacogenomics Research Network (PGRN)in 2009. CPIC® guidelines are indexed in PubMed as clinical guidelines, endorsed by ASHP and ASCPT, and referenced in ClinGen and PharmGKB.

Guidelines
CPIC® guidelines are designed to help clinicians understand HOW available genetic test results should be used to optimize drug therapy, rather than WHETHER tests should be ordered. A fundamental assumption underlying the CPIC® guidelines is that clinical high-throughput and pre-emptive (pre-prescription) genotyping will become more widespread and that clinicians will be faced with having patients’ genotypes available even if they have not explicitly ordered a test with a specific drug in mind. Several professional societies have endorsed CPIC®’s guidelines, processes and projects.
Each CPIC® guideline adheres to a standard format and includes a standard system for grading levels of evidence linking genotypes to phenotypes, how to assign phenotypes to clinical genotypes, prescribing recommendations based on genotype/phenotype, and a standard system for assigning strength to each prescribing recommendation.
Exploring CPIC® Guidelines
Visit the CPIC® Guidelines website. Under the Drugs column, select a drug you provide frequently in clinical practice and explore the associated genes and guidelines. For example, the CYP2D6 gene is common in several medication pathways, including those for opioids, SSRIs, TCAs, beta-blockers, and more. Variants in this gene can significantly impact how individuals metabolize many medications.
The PharmGKB YouTube channel has brief videos summarizing CPIC® guidelines for various drugs and genes.
Examples of Pharmacogenomics in Clinical Practice
Avoiding adverse drug effects
Healthcare professionals and researchers constantly seek to optimize medical treatments and avoid adverse reactions to treatments, which are estimated to affect between 7 percent and 14 percent of hospitalized patients. This makes adverse reactions a significant cause of added days spent in a hospital and the fourth leading cause of death in the United States.
Stevens-Johnson syndrome
One example of such an adverse reaction is Stevens-Johnson syndrome (SJS), a severe allergic reaction also called “scalded skin syndrome.” It can be caused by infections and very common medications like ibuprofen, anti-seizure medicines, or antibiotics. Patients may go from taking two pain pills to ending up in the hospital burn unit fighting for their lives if SJS progresses to a worse condition called toxic epidermal necrolysis (TEN). TEN is diagnosed when patients shed at least one-third of their skin off their bodies. Needless to say, anything we can do to prevent this allergic reaction is vitally important.
In Taiwan, married scientists Wen-Hung Chung (a physician) and Shuen-Iu Hung (an immunologist) noticed that SJS/TEN was much more common in patients taking carbamazepine, used to treat epilepsy and seizures, or allopurinol, used to treat gout. They showed that this was due to genomic variants in the HLA-B gene. Not surprisingly, this gene helps control the immune response. As a result of their work, the country of Thailand has implemented genomic testing before these medications are prescribed. The results of this “pharmacogenomic test” are used to decide whether it is safe to give a specific patient certain medicines, like carbamazepine or allopurinol. Thailand’s government even covers the cost of this testing, and the frequency of SJS/TEN has been drastically reduced. We have since learned that different ancestries are associated with different HLA-B genomic variants, so countries may need different approaches to monitor which medications are most likely to be linked to SJS/TEN.
Concept in Action
Watch Pharmacogenomic Testing – Karen’s story (3 mins) on YouTube for a personal story of Karen’s experience with SJS.
Video source: Mayo Clinic. (2017, May 3). Pharmacogenomic testing – Karen’s story [Video]. YouTube. https://www.youtube.com/watch?v=TVZVehYWLYw
Mercaptopurine
Although the field is still young, doctors already use pharmacogenomics to treat their patients. Acute lymphoblastic leukemia (ALL) is a type of cancer that mainly affects children and is often treated with the medicine mercaptopurine.
Children with certain genetic changes in the gene TPMT can have a severe reaction to this drug. Doctors will often test for these changes, and if they are present, will give the child a lower dose of mercaptopurine or use a different medicine.
Mercaptopurine belongs to a class of medicines called thiopurines. It is broken down in the body by several proteins, some of which convert mercaptopurine to active molecules that help kill cancer cells. Mercaptopurine is also broken down by a protein called TPMT, encoded for by the TPMT gene.
When TMPT breaks down mercaptopurine, it is inactivated. This inactivation prevents dangerous concentrations of active molecules from building up. Changes in the TPMT gene, such as the haplotypes *2, *3A or *3C, can reduce or stop the inactivation of mercaptopurine by the TPMT protein. This causes too many active molecules to build up in the body and can lead to a severe decrease in immune system activity, known as myelosuppression. Myelosuppression can be fatal.
Testing for TPMT variants is widely available: the Genetic Testing Registry provides over 20 laboratories currently offering this test. In Europe, testing before administration of thiopurines is becoming routine clinical practice – a survey in the United Kingdom found that 67% of clinicians ordered a TPMT test before prescribing azathioprine, another drug that is broken down by TPMT.
In the United Kingdom, testing for TPMT status is mandatory for children and young adults before treatment on the ALL2011 trial protocol, and the British Association of Dermatologists guidelines for the safe and effective prescribing of azathioprine states that TPMT activity should be checked in all patients before receiving azathioprine. The Clinical Pharmacogenetics Implementation Consortium (CPIC®) has also provided dosing guidance based on the TPMT genotype for thiopurines.
Tamoxifen
We have also learned that a person’s genome sequence is not everything regarding medication responses. The human body is a highly complex machine, and the instructions written in our DNA are just part of the process.
There are some cases, as with the breast cancer treatment tamoxifen, where a small study showed that there might be a relationship between someone’s response to the medicine and a variant in the CYP2D6 gene. However, this finding did not appear true in a larger study involving many more people. That’s why, at this time, the U.S. Food and Drug Administration (FDA) labelling for tamoxifen does not recommend CYP2D6 pharmacogenomic testing. However, the issue is still being reviewed as more research is conducted.
Another gene in the same CYP family, called CYP2C19, has variations which affect how your body can use clopidogrel (more commonly known as Plavix), an anticoagulant to prevent blood clots, and thus reduce the risk of stroke heart attack. If an individual has a variant causing malfunction of the CYP2C19 protein, they cannot process clopidogrel and either require a different dose or a different medication. As it turns out, these variants in CYP2C19 are also more common in those with Asian ancestry. Testing for variants in this gene is also not routinely recommended. However, individuals may wish to speak with their healthcare provider about the test if clopidogrel is prescribed, particularly if they have East Asian family members.
Pharmacokinetic and Pharmacodynamic Pathways
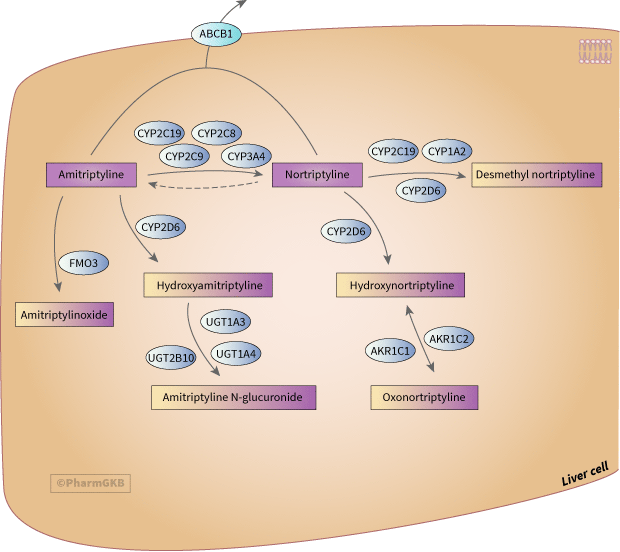
Background
Amitriptyline and nortriptyline are tricyclic antidepressants initially designed for use in the treatment of depression. Amitriptyline is also used to treat various types of pain, such as fibromyalgia and neuropathic pain [Article:15554244]. Nortriptyline is a metabolite of amitriptyline and a drug in its own right [Articles:15554244, 18359012]. Both drugs are non-selective monoamine reuptake inhibitors, preventing the reuptake of norepinephrine and serotonin at nerve terminals via interaction with their respective transporters, SLC6A2 and SLC6A4, and potentiating the action of these neurotransmitters. Additional effects and side effects occur due to cross-reactivity with opioid, cholinergic and adrenergic receptors [Articles:8736630, 10319193].
Metabolism
Amitriptyline and nortriptyline are readily absorbed in the GI tract and subject to extensive hepatic metabolism, with less than 5% of the drug eliminated unchanged (reviewed in [Article:10319193]). The main metabolizing enzymes with clinical significance for amitriptyline are CYP2C19 and CYP2D6 [Articles:23486447, 27997040]. CYP2C19 is the primary enzyme responsible for demethylation at physiological concentrations, while CYP2D6 carries out hydroxylation to less active metabolites [Articles:15554244, 18359012]. Hydroxynortriptyline is the most abundant metabolite of both amitriptyline and nortriptyline in humans [Article:10319193]. There are two enantiomers of hydroxynortriptyline and the E enantiomer is produced at a rate of around 5 times that of the Z enantiomer [Article:10319193]. Methylation of nortriptyline to amitriptyline has been reported in vivo in some case studies. A study that examined metabolite and prescription data from an extensive network of medical centers for whom amitriptyline measurements were available found that approximately 15% of patients receiving nortriptyline had significant levels of amitriptyline (above 28ng/ml) despite not having received the parent drug [Article:16553509]. The mechanism for methylation was not elucidated.
Pharmacogenomics
Many studies have examined variations in drug-metabolizing enzymes for their impact on amitriptyline and nortriptyline pharmacokinetics, with most focusing on CYP2D6 and CYP2C19. CPIC guidelines are available for both CYP2D6 and CYP2C19 and amitriptyline [Article:23486447]. Links to individual papers can be found under the PGx Research tab for amitriptyline and nortriptyline and clinical annotation summaries by haplotype under the Clinical PGx tab.
Attribution & References
Except where otherwise noted, this page is adapted from:
- Pharmacogenomics courtesy of National Human Genome Research Institute (NHGRI), Public Domain with attribution
- What is Pharmacogenomics? by PharmGKB, CC BY-SA 4.0
- What is CPIC®? by Clinical Pharmacogenetics Implementation Consortium, CC0 1.0 Universal (CC0 1.0) Public Domain Dedication.
CPIC guidelines and content are subject to updates and modifications, and users should refer to cpicpgx.org to confirm they are accessing the most current content - Amitriptyline and Nortriptyline Pathway, Pharmacokinetics by Carolyn F. Thorn, PharmGKB, CC BY-SA 4.0

