12.4 Genomics Applications by Specialty
While numerous practice areas are still progressing toward widespread integration of genomics into routine medical practices, certain specialties have seen more substantial advancements, often due to greater research funding or the strategic prioritization of genomics implementation. While the previous chapter focused on oncology as a leading example where the integration of genomics is well-established and extensively applied, key resources and emerging practices in each of these other five practice areas are provided to highlight the growing prevalence of genomics: pediatrics, maternity/obstetrics, mental health, neurology, and cardiology. It should be noted that the relevance of genomics is not limited to these practice areas.
Pediatrics
Units three and four briefly introduced a number of genetic conditions that manifest in childhood, along with early adverse experiences and their influence on the epigenome.
Recent advances in genomics mean that increasing numbers of children and their families can benefit from genomic testing. Testing can provide new diagnoses, shape management decisions, guide treatment options and provide valuable information.
Many genomic conditions affect early development, leading to congenital structural malformations and/or neurodevelopmental delay. This means that individuals with genomic conditions will commonly present in childhood.
Identifying a diagnosis can help patients and their families in a wide variety of ways. Some examples are outlined below.
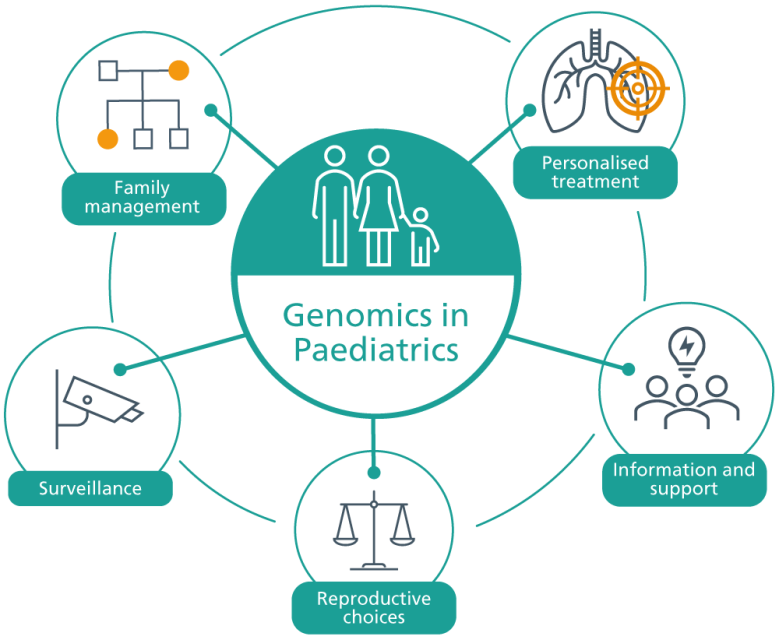
Information and support
Having a genomic diagnosis often allows the patient and their family – as well as other healthcare professionals – to learn more about their condition, understand the health implications in the short and long term, and access family support groups, which may be helpful from both an emotional and an educational perspective. Support groups often inform their members about developments in the field, including research and clinical trials.
Surveillance
Some pediatric genomic conditions are associated with specific health complications, such as an increased risk of developing early childhood tumours. For some of these complications, specific surveillance guidelines have been developed and published; this means that if the genomic condition is diagnosed early on, the child will have timely access to a condition-specific surveillance program that could improve their healthcare outcomes by identifying and enabling management of complications (which may include personalized treatments) at an early stage.
Personalized treatment
In some cases, a genomic diagnosis provides access to personalized treatment. Some examples of this are outlined below.
- The use of commonly-used drugs proven to be particularly effective in a specific genomic condition. For example, the anticonvulsant retigabine (ezogabine) has particular efficacy in treating epilepsy caused by loss-of-function variants in the KCNQ2 gene.
- The avoidance of certain medications. For example, some babies (1 in 500) are born with a particular genetic variant that means that administering the antibiotic gentamicin will cause irreversible hearing loss; however, new technologies have enabled clinicians to test for this genetic variant with a single cheek swab, which has a 15-minute turnaround time for results and means that use of gentamicin can be avoided where necessary.
- The use of targeted therapies – many of which are in development – such as enzyme replacements, dietary restrictions or gene therapies. For example, patients with spinal muscular atrophy are now benefiting from treatment with nusinersen (widely marketed as Spinraza), a drug that is delivered directly to the central nervous system to target an underlying cause of motor neuron degeneration.
Family management and reproductive choices
Because we share some of our DNA with people to whom we are genetically related, genomic conditions can have implications for our family members. This means that we must consider the inheritance pattern of any genomic diagnosis. It may be appropriate to offer parental testing, which helps to clarify any inheritance patterns and potential risks to immediate family members. For some conditions, it may be appropriate to offer cascade testing to the wider family.
Where there is a significant chance of future pregnancies being affected by the same genomic condition, some couples may wish to make complex, personal reproductive choices that may include a normal pregnancy (sometimes with additional scans or screening), use of donated sperm, egg and/or embryo, adoption, prenatal diagnosis or pre-implantation genetic diagnosis (PGD). Prenatal diagnosis is usually performed through chorionic villus sampling between 11 and 14 weeks of pregnancy. PGD is an IVF-based technique in which embryos are screened and only those without the familial genomic variant are transferred into the womb. In these scenarios, your role as a pediatrician would be to refer your patients to the appropriate specialist team – be it genetic counselling, fetal and women’s health or clinical genetics.
Finally, children with a genomic condition may be anxious about and want to discuss the implications of their diagnosis for any future children they themselves may have. An understanding of the inheritance patterns of genomic conditions will help you, as a pediatrician, to have those conversations with affected children.
The number of ways in which genomics can be applied in pediatrics will only increase with time. Take a look at the diagram below to find out some of the ways in which genomics is already being used to improve pediatric patient outcomes.
Case Example: Bardet-Biedl Syndrome
- Antenatally, George was found to have an echogenic kidney (a kidney that appears bright in an ultrasound, indicating a possible condition). He was also born with an extra fifth digit (a condition known as polydactyly), and was consequently referred to the clinical genetics team.
- When George was reviewed by the clinical genetics team as a baby, results of microarray testing and a postnatal renal ultrasound scan were both normal.
- George was followed up by the clinical genetics team until the age of two. There were no further suspicions of a genetic disorder, so he was discharged back to pediatric care, where he was later managed for developing obesity and mild learning difficulties.
- Due to evolving genomic technologies, George was then recruited into a research study into early-onset obesity by his pediatrician.
- Two years later, the research study found two variants in George’s BBS5 gene, indicating a diagnosis of autosomal recessive Bardet-Biedl syndrome (BBS).
- As well as presenting a cause for George’s polydactyly, obesity and learning difficulties, this new diagnosis had important implications for his management and prognosis. Sadly, most individuals with BBS become blind by their second or third decade of life.
- George was referred back to the clinical genetics team for further support and counselling about the condition, while his pediatrician made clinical referrals to the ophthalmology, audiology, endocrinology and renal teams for surveillance of his syndromic manifestations.
Maternity
Genomics will continue to play more of a role in the diagnosis, management and treatment pathways for parents and their babies. Expectant parents need nurses who are knowledgeable, confident and competent so it’s paramount to ensure your genetics and genomics knowledge and skills are up to date with the latest developments. This will allow them to make choices with conviction and courage. Information is easier than ever to access online and people are now more aware about genomics – they could even know something that the nurse does not. Be prepared for questions and know where you can find accurate answers.
There is some overlap between maternity and pediatric specialties, depending on when a genetic condition is suspected or diagnosed. Nurses may care for patients at any point in their family planning or pregnancy journey, depending on their context of practice. Pre-natal, antenatal, and postnatal screening was briefly covered in unit eight. This will not be revisited here except to mention that it is nurses that typically obtain specimens for genetic screening in these contexts. To provide appropriate informed consent, nurses must know what the tests might reveal and the risks and benefits of testing.
Some types of tests are indeed genetic, because they test for a condition by looking for a variation in a specific gene. Genomics, however, describes how we interpret and act on information from the whole genome, or any part of it. The National Genomic Test Directory refers to all tests as ‘genomic’ and we will do the same throughout this resource.
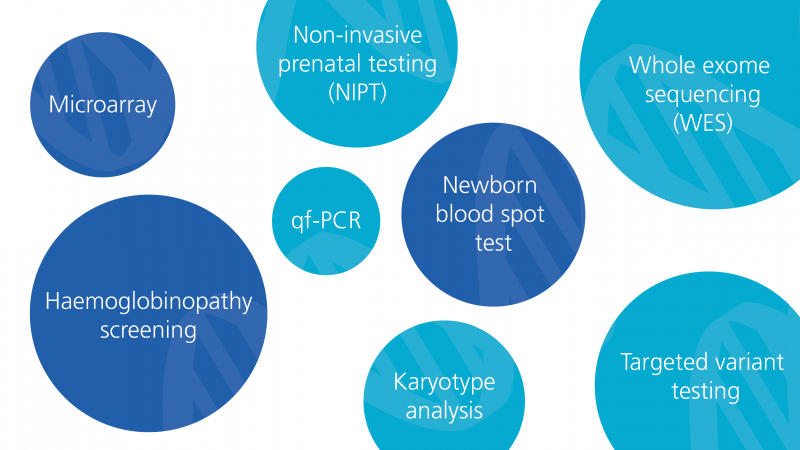
Look at the graphic to see the names of some genomic tests used in family planning or pregnancy. There is more information available here.
Our understanding of genomic information is increasing all the time. In addition, family histories change, and new information may alter clinical management or genetic risk assessment. If an expectant parent says something like “no-one was interested about this in my last pregnancy,” consider revisiting this information – there could be cause for referral to clinical genetics for further testing. It is also always worth taking another look at a family history, especially if the previous genomics consultation was several years ago.

Referrals for genomic counselling or testing should be made at the earliest possible opportunity as the test results can provide information that can have a considerable impact on care. Some genomic tests can take several weeks to come back, so a fast referral is crucial. It is important to ‘think genomics’ whenever you ask about a personal, obstetric and family history, and to take timely, appropriate action if you notice a red flag.
Nurses working in labour and delivery may experience some of the following circumstances that require genomic literacy:
- A nurse could be the first to notice signs and symptoms that may indicate the baby has a genetic condition.
- A nurse could be involved in the delivery of a baby that was diagnosed with a genetic condition during the pregnancy or that has been identified as having a higher chance of having a condition, based on a screening test result or because of the family history.
- Nurses might be caring for an expectant parent who has a genetic condition themselves, meaning they are at higher risk during labour.
| Condition | How it can affect the pregnancy |
|---|---|
| Loeys-Dietz or vascular Ehlers Danlos syndrome | Increased risk of uterine/vascular rupture. |
| Cystic fibrosis or sickle cell disease | Increased risk of complications around labour. |
| MCADD | Dietary considerations. Also has a considerable impact on neonatal care for the baby after birth (see our case study: Sara’s story). |
| Haemophilia (carrier) | Increased risk of port-partum haemorrhage due to changes in factor VIII levels. |
| Marfan syndrome | Increased risk of complications around labour |
| Epidermolysis bullosa (EB) | Potential for huge changes in care and management, both during the pregnancy and neonatally. Read more in this document: ‘Epidermolysis bullosa (EB) – information for pregnancy and childbirth’ [PDF] |
| Factor V Leiden | Increased risk of clotting disorders in pregnancy, and of miscarriage. Autosomal dominant condition. |
- Nurses could be involved in the delivery of a baby following termination of pregnancy, or a stillborn baby due to genomic complications.
Mental health
The challenge of the mind
Throughout the history of psychiatry, researchers have tried but failed to find any physical basis for the strikingly abnormal experiences and behaviours of patients with severe mental illness.
There are no blood tests or brain scans that can help us understand the nature of mental illness. Diagnoses are based on patterns in the clinical presentation, such as mood changes, delusions or ritualistic checking, and medications are prescribed on an empirical basis: we know that they work (sometimes with a surprising level of effectiveness), but we don’t really know why.
Mental illnesses are especially difficult to research because invasive investigations of the brain are not possible in the way they are for other organs. Even when physiological differences can be measured, it is often impossible to distinguish whether these are causes or effects of mental illness.
Hope through research
It’s now believed that our genes could well play a part in our susceptibility to any given illness, and the effect of genetics on the risk of schizophrenia and bipolar disorder are well evidenced and substantial.
Genomic research offers the hope of better understanding the root causes of mental illness: by finding specific genes which are involved in these devastating illnesses, we might gain some understanding of the pathological processes leading to their development and ultimately develop better treatments.
CNVs in schizophrenia
The discovery with the most immediate clinical relevance relates to schizophrenia. Research has found that some patients with the condition have a chromosomal abnormality called a copy number variant (CNV) – where there is either an extra copy of part of the chromosome (a duplication) or a part that is missing completely (a deletion).
There are around 12 known locations on the chromosome where a CNV results in a substantially increased risk of schizophrenia – perhaps a thirty-fold or more increase above the background risk of 1%. If such a CNV is present in a person with schizophrenia, then it would be reasonable to say that the CNV had ‘caused’ the illness.
Wider implications
Typically, a CNV will impact several different genes and it has proved difficult to identify which of these are specifically responsible for increasing risk. Nevertheless, discovering that a patient carries a CNV has important implications:
- Validation: Finding a CNV can provide the patient and those around them with a clear, concrete explanation for why they have become unwell. For many people, it can be difficult to accept that mental illness is real, and finding a definitive cause can help the person to understand and accept that their condition is valid and deserving of treatment.
- Associated conditions: As well as being the primary cause of a condition like schizophrenia, some CNVs are also associated with increased risk of additional physical health conditions. Finding a CNV therefore presents an opportunity to investigate, and possibly discover, an illness that was previously undiagnosed.
- Impact on family: Any genetic diagnosis can also have implications for family members, and younger siblings of patients with schizophrenia often express anxiety that they may also develop the illness. If the patient carries a CNV but the sibling does not, then they can be reassured that they are at no increased risk.
The proportion of people with schizophrenia in which a CNV is found is small – around 2% – but since the identification of a CNV has important implications for them and their family, and since the test is simple and inexpensive, some psychiatrists argue that testing for CNVs should be routine for those with a new diagnosis.
Individual genes
It is known that when the function of certain genes is disrupted by variants in a person’s DNA, the risk of schizophrenia can rise. However, implicating individual genes has proved challenging, and the first to be identified, SETD1A, did not provide insights into how its disruption might lead to schizophrenia, as had been hoped.
In 2019, research by an international collaboration called SCHEMA, analyzed thousands of exomes, and identified a handful of genes where loss of function substantially affects schizophrenia risk – with some genes appearing to be linked to disease processes.
The clearest example relates to a particular receptor molecule for the neurotransmitter glutamate. It was already known that drugs blocking this receptor, as well as a syndrome called autoimmune encephalitis where antibodies attack the receptor, could cause symptoms that are similar to schizophrenia. Researchers have now demonstrated that variants that disrupt a gene coding for this receptor also increase schizophrenia risk. Thus, pharmacological, immune and genetic results all suggest that impaired functioning of this glutamate receptor can increase the risk of developing psychotic symptoms and other problems associated with schizophrenia.
These findings offer an opportunity to try to develop better drug treatments which might modify receptor functioning and lessen symptoms.
Where we are now
Psychiatric genomics is a burgeoning area of research and ever-expanding clinical application. For example, the Psychiatric Genomics Consortium is a collaboration of over 800 international scientists working with the pooled data of almost 1 million participants to uncover the role of genetics in psychiatric disorders. Some other fascinating areas of implementation in this practice context include:
PharmGKB provides evidence-based information and CPIC guidelines on pharmacogenomics for drugs, genes, and phenotypes involved in substance use disorder.
In this recent study published in November 2024, researchers used machine learning (AI) to detect complex structural variations in the genome that likely contribute to psychiatric disease.
The Mayo Clinic has a Psychiatric Genomics and Pharmacogenomics Program that focuses on bipolar disorder, alcohol use disorder, and psychiatric pharmacogenomics.
Discoveries of genetic causes of disordered eating has led to advances in treatment and hope for patients who have been unsuccessful with more traditional therapies.
Neurology
Rapid advances in technology and understanding mean that genomic testing is becoming much more integral to the field of neurology. Understanding the control of gene expression in the brain is central to understanding normal brain function, and increasingly neurological disease.
As genomic technology is enabling progress in our understanding of the etiology of disease, it is enabling the development of new targeted therapies. Rapid progress in clinical trials and drug development are expected over the next few years. There are now trails underway for Parkinson’s disease and motor neurone disease, for example, targeting specific genes such as LRRK2 and C9orf72.
How is genomics used in neurology?

Diagnosis
Genomic testing is particularly useful in the diagnosis of familial and early-onset neurodegenerative diseases, for example ataxia, hereditary spastic paraparesis and some forms of dementia and Parkinson’s. Disorders such as epilepsy can have a single genetic variant in about 30% of affected individuals (Jain et al., 2019), and more is becoming known about the genetics of neurodevelopmental disorders such as autism (Aldridge et al., 2024).
Predictive testing
Genomic test results can impact whole families. If variants are found early, it can enable better clinical management for the whole family and enable individuals to consider their options for the future. For example, identification of the Huntington’s gene variant in a family with Huntington disease enables the counselling of other family members for risk-management including possible predictive testing and referral to relevant clinical trials.
Targeted treatment
In some instances, a precise genetic diagnosis means an individual can access gene-directed therapies which have a good chance of benefiting them.
Case Example: SMA
An example is the treatment of spinal muscular atrophy (SMA) with Nusinersin. It is hoped that other gene-directed therapies will soon be available.
- SMA is a rare, severely life-limiting and ultimately fatal neuromuscular condition which causes most affected children to die from respiratory failure within the first three years of life.
- Until 2016, there was no treatment for SMA and management in severe cases was with palliative care.
- SMA is caused by variants in the SMN gene, leading to loss of the SMN1 protein.
- In 2016, the therapy Nusinersin was introduced. Nusinersin works by upregulating a ‘back-up’ copy of the gene, SMN2.
- As a result of the availability of this therapy, children with a diagnosis who previously would have had a very limited life expectancy are able to lead much more normal lives.
- Although the long term effects of the treatment are not known, it is clear that it has a dramatic effect on both quality of life and life expectancy for affected children.
It is hoped that many more treatments, based on similar mechanisms, will be developed now that wider access to genomic technology and genomic data is possible.
Clinical trails and drug development
The identification of the genetic cause(s) of a disease makes it easier to develop targeted therapies. For example:
- The identification of variants in the synuclein gene in Parkinson’s disease, and identifications and variants in the APP and presenilin genes in Alzheimer’s disease, have allowed for the development of animal models. These animal models have been used to develop antibody-based treatments for these diseases, and those antibody treatments are now in clinical trials. Updates can be found on the Alzoforum website.
- Several years ago, the gene most commonly associated with motor neurone disease – the C9orf72 gene – was identified. A gene therapy trial is now in progress, looking to turn off the abnormal gene and potentially provide a treatment for affected patients (see ‘Genomics in Practice’ example below).
The widespread availability and relative accessibility of genomic testing will result in more trials, more patients being eligible for and recruited to trials, and, as a result, more rapid progress in the area of targeted treatments.
Cardiology
The field of cardiology is increasingly influenced by advances in genomics, which can be used in diagnosis, treatment and management in a number of ways. Health professionals in cardiovascular medicine are increasingly likely to encounter genetic and genomic information and should be aware of how to deal with it.
As well as a role in rarer, inherited cardiac conditions, genomics is increasingly going to have a role in assessment and management of common cardiovascular diseases such as hypertension and coronary disease.
Genomic research directly identifies genes and pathways underpinning disease that may represent new therapeutic targets. Genetics and genomics also have an important and growing role in patient stratification.
Families with inherited cardiac conditions (ICC)
Most ICCs are autosomal dominant, meaning that immediate family members have a 50% chance of inheriting the same condition. Cascade testing can be used to determine the risk to the patient’s family members.
Treatment and management of cardiac conditions
For some inherited cardiac conditions, treatment can be refined when we understand the precise molecular basis of an individual’s condition. For example, those with inherited arrhythmia may receive treatment tailored to the genetic cause.
Increasingly, other cardiological treatment can be chosen in accordance with an individual’s genetics. For example, genetic testing can determine whether an individual will be resistant to clopidogrel, or has an elevated risk of developing statin myopathy.
Genetic information can be used to intervene early. For example, adopting a favourable lifestyle has been shown to reduce coronary disease risk by around 50% even in the presence of a high genetic predisposition.
To read some examples of Canadian nurses conducting research on the psychosocial aspects of participating in predictive genetic testing for cardiovascular conditions or living with autosomal dominant cardiac conditions, read Manuel and Brunger (2015) and Manuel and Brunger (2014).
New therapies
Understanding the genetic basis of disease can give us an insight into the molecular mechanisms and pathways involved, which can allow us to develop new treatment strategies. For example, specific disease-modifying targeted therapies are in phase 3 trials in both dilated and hypertrophic cardiomyopathies.
Inherited conditions can serve as genetic models for more common forms of disease. For example, PCSK9 inhibitors – a new class of lipid lowering therapy – were developed as a result of studies into familial hypercholesterolaemia.
New genome technologies are also offering the promise of new therapies through gene repair or replacement. Additional resources for cardiogenomics include GECKO and the University of Ottawa Heart Clinic.
Attribution & References
Except where otherwise noted, this page is adapted from:
- Pediatrics section adapted from Genomics in Paediatrics by Genomics Education Programme, CC BY-NC 4.0
- Maternity section adapted from Genomics in Midwifery by Genomics Education Programme, CC BY-NC 4.0
- Mental health section adapted from Keeping genes in mind: genomics and mental illness by Genomics Education Programme, CC BY-NC 4.0
- Neurology section adapted from Genomics in Neurology by Genomics Education Programme, CC BY-NC 4.0
- Cardiology section adapted from Genomics in Cardiology by Genomics Education Programme, CC BY-NC 4.0
References
Aldridge, C., Morrison, S., Allanson, J. E., WAlji, S., & J. C. Carroll. (2024, May). Neurodevelopmental disorders. Genomics Education Canada: Knowledge Organization (GECKO). https://www.geneticseducation.ca/resources-for-clinicians/neurogenomics/ndd-main-landing/ndd-otr
Jain, P., Dyment, D., & Snead, O. c. (2019, July). GECKO on the run: Genetics of epilepsy. Genomics Education Canada: Knowledge Organization (GECKO). https://www.geneticseducation.ca/uploads/GECKO%20On%20the%20Run%20-%20Epilepsy%20-%20Final%20-%20Jan2019.pdf
Manuel, A., & Brunger, F. (2014). Making the decision to participate in predictive genetic testing for arrhythmogenic right ventricular cardiomyopathy. Journal of Genetic Counseling, 23(6), 1045–1055. https://doi.org/10.1007/s10897-014-9733-4
Manuel, A., & Brunger, F. (2015). “Awakening to” a new meaning of being at-risk for arrhythmogenic right ventricular cardiomyopathy: a grounded theory study. Journal of Community Genetics, 6(2), 167–175. https://doi.org/10.1007/s12687-015-0212-x

