Ontogeny refers to an organism’s development across its entire lifecycle, from fertilization to adulthood. During this process, the genetic blueprint of the organism is expressed under the influence of external factors. In cases of nutrient deficiency, essential organs like the brain receive the majority of the scarce nutrients, prioritizing them over less critical organs, such as the pancreas or kidneys. Research has indicated that if such nutrient allocation happens during a critical developmental period, changes in cellular structure and function may become permanent.
3.5 The Exposome
Epigenetics and the Influence of the Environment on Gene Expression – The Exposome
Ontogeny
Epigenetics and age
Your epigenetics change throughout your life. Your epigenetics at birth are not the same as your epigenetics during childhood or adulthood.
EXAMPLE: A newborn, 26-year-old, and 103-year-old. Scientists measured DNA methylation at millions of sites in a newborn, 26-year-old, and 103-year-old. The level of DNA methylation decreased with age. The newborn had the highest level of DNA methylation, the 103-year-old had the lowest level of DNA methylation, and the 26-year-old had a DNA methylation level that was between that of the newborn and the 103-year-old (Heyn et al., 2012).
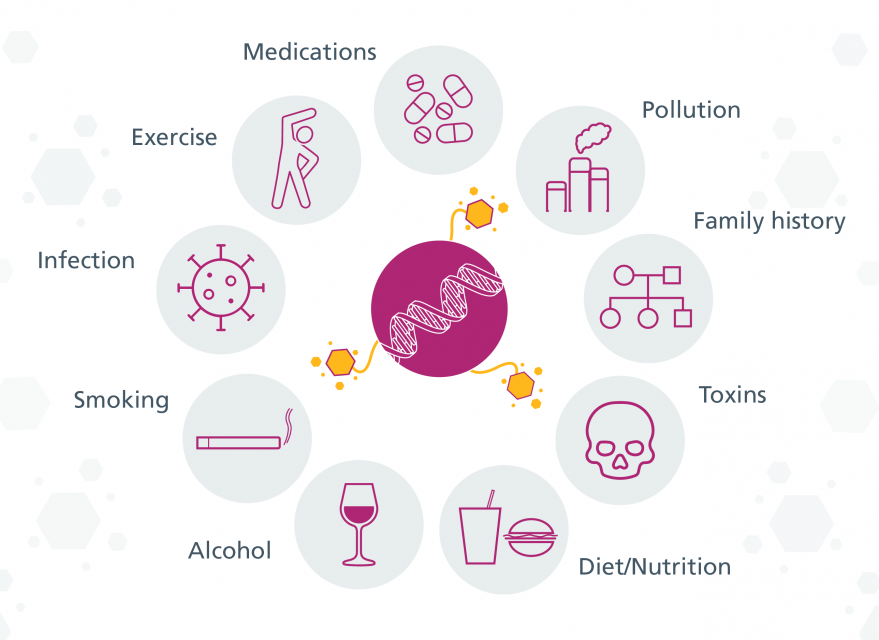
The Exposome
Success in mapping the human genome has fostered the complementary concept of the “exposome”. The exposome can be defined as the measure of all the exposures of an individual in a lifetime and how those exposures relate to health. An individual’s exposure begins before birth and includes insults from environmental factors and occupational sources. Understanding how exposures from our environment, diet, lifestyle, etc. interact with our own unique characteristics such as genetics, physiology, and epigenetics impact our health is how the exposome will be articulated.
Epigenetics and exposures
Your epigenetics can change in response to your behaviors and environment.
Factors that influence the epigenome
Most epigenetic modifications are transient and reversible, allowing our cells to respond and adapt to changes in environment and behaviour. Although they happen on a molecular level, they can have a considerable impact on us. External factors, such as diet and lifestyle can also influence them.
Nutrition during pregnancy
A pregnant person’s environment and behavior during pregnancy, such as whether they eat healthy food, can change the baby’s epigenetics. Some of these changes can remain for decades and might make the child more likely to get certain diseases.
EXAMPLE: Dutch Hunger Winter famine (1944–1945). People whose mothers were pregnant with them during the famine were more likely to develop certain diseases, such as heart disease, schizophrenia, and type 2 diabetes (Roseboom, 2019). Around 60 years after the famine, researchers looked at DNA methylation levels in people whose mothers were pregnant with them during the famine. These people had increased DNA methylation at some genes and decreased DNA methylation at other genes, compared with their siblings who were not exposed to famine before birth (Tobi et al., 2018). These differences in DNA methylation could help explain why these people had an increased likelihood for certain diseases later in life (Roseboom, 2019; Tobi et al., 2018; Dayeh et al., 2016).
Concept in Action – Maternal Nutrition
Watch this short video to see how animals models contributed to this theory of maternal nutrition. How can two genetically identical mice look completely different?
Watch Why do Two Genetically Identical Mice Look Vastly Different? (3 mins) on YouTube
Video source: World Science Festival. (2013, November 22). Why do two genetically identical mice look vastly different? [Video]. YouTube. https://youtu.be/IYJ_nd9glvw
Smoking
Exposures such as smoking can cause epigenetic changes. However, these epigenetic changes can be reversible in some cases.
EXAMPLE: Smokers, nonsmokers, and former smokers. Smoking can result in epigenetic changes. For example, at certain parts of the AHRR gene, smokers tend to have less DNA methylation than nonsmokers. The difference is greater for heavy smokers and long-term smokers. After quitting smoking, former smokers can begin to have increased DNA methylation at this gene. Eventually, they can reach levels similar to those of nonsmokers. In some cases, this can happen in less than a year, but the length of time depends on how long and how much someone smoked before quitting (McCartney et al., 2018).
Concept in Action – Foundational Studies
This video provides a good overview of some of the foundational studies that form the basis of what we know about epigenetics today. It also illustrates the many ways in which the exposome can impact the epigenome.
Watch Epigenetics with Dr. Moshe Szyf (Part 1) (18 mins) on YouTube
Video source: CGTN America. (2020, January 11). Epigenetics with Dr. Moshe Szyf (part 1) [Video]. YouTube. https://www.youtube.com/watch?v=OEAJmDPJz_I
Food for thought: Consider the legacy of generational trauma resulting from colonialism. What connections can you make between intergenerational trauma and some of the epidemiological patterns you may be familiar with regarding non-communicable diseases in some of the populations affected by colonialization? What role does epigenetics play in these health outcomes?
Exposomics
Exposomics is the study of the exposome and relies on the application of internal and external exposure assessment methods. Internal exposure relies on fields of study such as genomics, metabonomics, lipidomics, transcriptomics and proteomics. Commonalities of these fields include 1) use of biomarkers to determine exposure, effect of exposure, disease progression, and susceptibility factors, 2) use of technologies that result in large amounts of data and 3) use of data mining techniques to find statistical associations between exposures, effect of exposures, and other factors such as genetics with disease. External exposure assessment relies on measuring environmental stressors. Common approaches include using direct reading instruments, laboratory-based analysis, and survey instruments. The extent to which internal and external exposure assessment will contribute to our understanding of the exposome is being debated as each approach has certain merits.
A key factor in describing the exposome is the ability to accurately measure exposures and effect of exposures. Many of the “omics” technologies have the potential to further our understanding of disease causation and progression. Metabonomics and adductomics (DNA and protein adduct measurement) have been used in the past to establish exposure-disease relationships. Research is needed to determine the utility of the “omics” technologies in defining the exposome.
Why should we study the exposome?
One of the promises of the human genome project was that it could revolutionize our understanding of the underlying causes of disease and aid in the development of preventions and cures for more diseases. However, genetics has been found to account for only about 10% of diseases, and the remaining causes appear to be from environmental causes. So to understand the causes and eventually the prevention of disease, environmental causes need to be studied.
What are the challenges of advancing exposomics?
Some challenges that may limit the progress in this field of study are evident. An individual’s exposome is highly variable and dynamic throughout their lifetime. The impact of exposures can also vary with the individual’s stage of life. For examples, exposure to the drugs thalidomide or valproic acid during specific developmental periods in utero causes malformation of limbs; exposure to lead in infants and early childhood can lead to cognitive deficiencies. Exposures during early years may also predispose an individual to certain chronic diseases later in life.
The impact of environmental or occupational exposures can be different for each individual because of differences in genetic and other personal factors. Some people will develop a disease while another person with the same or greater exposure will not. The exposome may help to determine the underlying causes for this difference. Mapping an entire exposome for an individual will be difficult, if not impossible because of the complexity of a life-time of exposure. Specific exposures can be difficult to measure due to lack of sensitive methods or not knowing that an exposure has even occurred. Even when the exposure is known, measuring that exposure can be difficult since the indicators of exposure may be transient, such as for most chemicals, which are rapidly excreted and only a short time frame exists to directly measure them. In other cases, past exposure can be defined using legacy biomarkers. A common example of a legacy biomarker is antibodies produced by exposures to environmental or occupational insults.
The experience in studying genetic involvement in diseases serves as a model for studying the relationship between exposures and disease. In the past, hypotheses of the role of specific genes in disease were tested. Currently, genome-wide association studies are performed with the aid of new technologies which produce cheaper and faster analyses to generate hypotheses about the relationship between genes and disease. These studies identify gene pathways associated with disease, and, when an association has been identified specific hypotheses about the role of specific genes in disease can be generated and tested. An approach to the exposome is to use internal biological media and measure multiple endpoints. The data would be analyzed to identify associations between health outcomes and biomarkers of exposures, biomarkers of response, or patterns of biomarkers (exposure-wide association studies).
One important aspect of the exposome will be adherence to strict ethical principles as the exposome is deciphered. This will be paramount to ensure that the rights of individuals are not compromised when determining exposures and the relationship to their health.
Attribution & References
Except where otherwise noted, content on this page has been combined and adapted from:
- Epigenetics In Talking Glossary of Genomic and Genetic Terms by National Human Genome Research Institute, Public Domain
- Epigenetics by Health Education England’s Genomics Education Programme (GEP), CC BY-NC 4.0
- What is Epigenetics? by Health Education England’s Genomics Education Programme (GEP), CC BY-NC 4.0
- Epigenetics, Health, and Disease In Genomics and Your Health by CDC, Public Domain with attribution
References from original sources
Dayeh, T., Tuomi, T., Almgren, P., Perfilyev, A., Jansson, P. A., de Mello, V. D., Pihlajamäki, J., Vaag, A., Groop, L., Nilsson, E., & Ling, C. (2016). DNA methylation of loci within ABCG1 and PHOSPHO1 in blood DNA is associated with future type 2 diabetes risk. Epigenetics, 11(7), 482–488. https://doi.org/10.1080/15592294.2016.1178418
Heyn, H., Li, N., Ferreira, H. J., Moran, S., Pisano, D. G., Gomez, A., Diez, J., Sanchez-Mut, J. V., Setien, F., Carmona, F. J., Puca, A. A., Sayols, S., Pujana, M. A., Serra-Musach, J., Iglesias-Platas, I., Formiga, F., Fernandez, A. F., Fraga, M. F., Heath, S. C., Valencia, A., … Esteller, M. (2012). Distinct DNA methylomes of newborns and centenarians. Proceedings of the National Academy of Sciences of the United States of America, 109(26), 10522–10527. https://doi.org/10.1073/pnas.1120658109
McCartney, D. L., Stevenson, A. J., Hillary, R. F., Walker, R. M., Bermingham, M. L., Morris, S. W., Clarke, T. K., Campbell, A., Murray, A. D., Whalley, H. C., Porteous, D. J., Visscher, P. M., McIntosh, A. M., Evans, K. L., Deary, I. J., & Marioni, R. E. (2018). Epigenetic signatures of starting and stopping smoking. EBioMedicine, 37, 214–220. https://doi.org/10.1016/j.ebiom.2018.10.051
Roseboom T. J. (2019). Epidemiological evidence for the developmental origins of health and disease: effects of prenatal undernutrition in humans. The Journal of endocrinology, 242(1), T135–T144. https://doi.org/10.1530/JOE-18-0683
Tobi, E. W., Slieker, R. C., Luijk, R., Dekkers, K. F., Stein, A. D., Xu, K. M., Biobank-based Integrative Omics Studies Consortium, Slagboom, P. E., van Zwet, E. W., Lumey, L. H., & Heijmans, B. T. (2018). DNA methylation as a mediator of the association between prenatal adversity and risk factors for metabolic disease in adulthood. Science advances, 4(1), eaao4364. https://doi.org/10.1126/sciadv.aao4364

