12.3 Cancer Genomics
Proto-oncogenes
Recall from chapter 2.4 that control of cell division involves many different genes. Some of these genes act as signaling molecules to activate normal progression through the cell cycle. Think of oncogenes as the “accelerators” of cell growth and proliferation. One of the pre-requisites for cancer occurs when one or more of these activators of cell division become altered. Proto-oncogenes are normal genes that, when altered, become oncogenes and contribute to cancer as they code for positive cell-cycle regulators.
Tumour Suppressor Genes
Tumor suppressor genes act like the “brakes” of the cell cycle, preventing uncontrolled growth and promoting repair. More than 30 genes, including BRCA1 and p53, are classified as tumor suppressors. These genes repair DNA, induce apoptosis, and prevent abnormal cell division. Loss-of-function variants in these genes contribute to cancer progression, and both alleles must be varied (loss of heterozygosity) for abnormal growth to proceed.
Why is genomics important in cancer care?
Cancer is a disease of the genome. It occurs when variants in the genome result in uncontrolled cell growth and division. These genomic variants can be inherited from a parent or acquired at some point during a person’s lifetime. Most cancers are caused by acquired genomic variants. In around 5%-10% of cases, the individual has inherited a variant that greatly increases their chances of developing cancer.
Cancer care has rapidly evolved, particularly over the last 15-20 years. View genomics milestones in oncology using this Genomic Milestones interactive site.
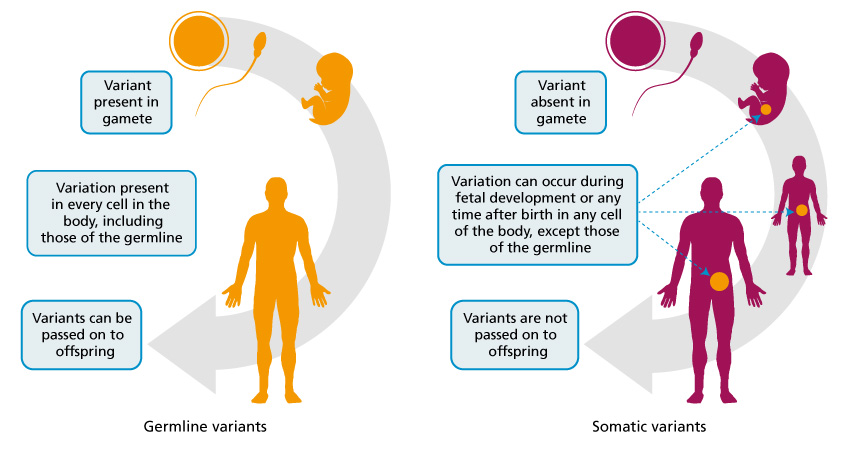
Two kinds of cancer variants
Inherited variants
Inherited genomic variants are also called germline variants. For an individual with a cancer predisposing germline variant (i.e. known to be pathogenic or likely pathogenic) each cell in their body already has an altered copy (see ch. 8.4 – Interpreting Genetic Tests) . This significantly increases their lifetime risk of cancer.
A patient with this type of variant may be offered additional screening or prophylactic surgery. For example, patients with particular BRCA1 and BRCA2 gene variants that are known to be pathogenic or likely pathogenic may opt for enhanced breast screening protocols, preventative medications, and reproductive health planning and counselling. An additional option is to have a preventative mastectomy or oophorectomy.
It is also important to consider the implications for the patient’s family, as appropriate testing (cascade testing) can identify other at-risk relatives who may be able to take measures to reduce their chance of developing cancer. Therefore, not only do nurses need to consider the autonomy of their patient, but their relational autonomy, including their networks and relationships that factor into their decision-making.
Acquired variants
Acquired genomic variants are called somatic variants, and these variants are present only in cancer cells. These variants are not inherited and cannot be passed on to any children.
Somatic variants can be the result of exposure to environmental factors, such as ultraviolet light, smoking, radiation and alcohol, or they can be entirely random. Each time a cell divides, errors might be introduced. While there are many mechanisms within the cell to correct these errors, occasionally they are missed.
Is cancer hereditary?
The term hereditary cancer is a bit misleading. It is important to note that not all cancers are hereditary and for those that are, cancer itself cannot be inherited. Specifically, somatic variants in tumour cells are not passed down to offspring. However, an inherited germline genetic variant increases the risk of developing certain types of cancer because at birth they have inherited one altered “copy” of the cancer causing gene. Inheriting a variant does not guarantee that person will develop cancer. However, their risk is increased over those without the specific variant for particular types of cancer.
For example, if a parent passes a BRCA1 or BRCA2 gene variant to their child, the child will have a much higher risk of developing breast and several other cancers. That’s why cancer sometimes appears to run in families.
Watch Genomics in Medical Specialties – Oncology: Cancer Treatment (3 mins) on YouTube
Video source: Genomics Education Programme. (2018, December 16). Genomics in medical specialties – Oncology: Cancer treatment [Video]. YouTube. https://www.youtube.com/watch?v=Putm4DHuj84
Blood Tests to Detect Cancer
Did you know that we are increasingly able to detect cancers by testing just a blood sample? Or that we are moving toward treating cancers not by where they are found in the body, but by how their genomes have changed? Cancer is caused by changes in an individual’s genome, but advances in DNA sequencing technology are leading to a new understanding of cancer and new ways for diagnosing and treating many types of cancer.
Cancer is a group of genetic diseases that result from changes in the genome of cells in the body, leading them to grow uncontrollably. These changes involve DNA variants in the genome. Our cells are constantly finding and fixing variants that occur in our genome as the cells divide over and over again. But on rare occasions, some variants slip through our cells’ repair machinery, and those variants can lead to cancer. The Human Genome Project has allowed us to establish what “normal” usually looks like for a human genome, so that we can now tell when changes in our genome have taken place that lead to cancer.
Large projects around the world, like The Cancer Genome Atlas in the United States and the Catalogue of Somatic Mutations (COSMIC) in the United Kingdom, have now determined the genome sequences of thousands of cancer samples of many cancer types. These projects have shown that some cancers have variants in the same group of genes, even though they may have started in completely different tissues. Many of the variants activate genes that normally promote cell growth or break genes that normally prevent cell growth. If we know more about the specific variants that led to someone’s cancer, no matter what tissue it was located in, then we can look for more specific and effective treatments for their cancer.
Unfortunately, some cancers are harder to evaluate because looking at their genomes would require difficult and painful biopsies or operations where tiny parts of the cancer tissue are removed for study. This also makes it harder for clinicians to monitor how treatment is working for some cancers because repeated biopsies are just not possible. Recent breakthroughs now allow the detection of circulating tumour DNA (or ctDNA) in the blood of patients instead of directly sampling the tumour. As cancer cells grow very fast and die, they release some of their DNA into the bloodstream. We now have tests that are sensitive enough to detect and sequence these pieces of ctDNA in the bloodstream separately from the normal DNA of the patient – this is called a “liquid biopsy.”
Although liquid biopsies are not yet in widespread use for cancer detection, improvements are being made all the time that move us closer to the routine use of ctDNA tests. One of the current approved uses for ctDNA is to test progression of non-small cell lung cancer by looking for specific variants in the EGFR gene over time. Liquid biopsies can point to who will likely relapse after treatment, by detecting DNA with EGFR variants that is circulating in the blood, sometimes better and more quickly than the now-used standard imaging techniques. The CHARM study is ongoing in five Canadian provinces. Visit their website cfDNA in Hereditary and High-Risk Malignancies (CHARM) study. This is an excellent example of research revolutionizing how hereditary cancer syndromes can be managed in practice. Their aim is to to develop a blood test to predict cancer development in carriers of cancer predisposition genes including BRCA1, BRCA2, PALB2 (hereditary breast and ovarian cancer) CDH1 (hereditary diffuse gastric cancer) MLH1, MSH2, MSH6, PMS2, EPCAM (Lynch syndrome) TP53 (Li-Fraumeni syndrome) using circulating tumour DNA. On an international level, the CASCADE study aims to provide support and care coordination to families with pathogenic variants connected to breast and ovarian cancer and to Lynch syndrome.
Refining treatment
Some genomic variants within the cancer genome can be used to work out the most appropriate treatment for the patient. Some variants can make the person more, or less, likely to respond well to particular treatments.
For example, tumours with certain variants in the EGFR gene respond well to EGFR-inhibitor drugs, but those without such variants do not. So two people with the same diagnosis of breast cancer may have different treatments based on the genomic information from their tumour.
Novel treatments can also be identified by sequencing the tumour’s genome.
Listen to specialist registrar Dr Alison Berner discuss the impact this is having on patients. Watch The Realisation of Personalised Medicine (3 mins) on Vimeo.
Video source: Genomics Education Programme. (2019). The realisation of personalised medicine [Video]. Vimeo. https://vimeo.com/336816796
By far, most cancers are not inherited, but there are a few examples of inherited cancers like Lynch syndrome (also known as hereditary non-polyposis colorectal cancer). This disorder is due to inherited variants in any of five different genes, and leads to an increased risk of different types of cancers, most often in the colon. Breast cancer is another example; again most cases are not inherited, but men or women who have inherited variants in the BRCA1 or BRCA2 genes have a much higher chance for developing breast cancer than other people.
| High penetrance breast and/or gynecologic cancer susceptibility genes | Moderate-penetrance genes associated with breast and/or gynecologic cancers | |
|---|---|---|
| BRCA1 and BRCA2 | Fanconi anemia genes | |
| Li-Fraumeni Syndrome (LFS) | ATM | |
| PTEN Hamartoma Tumor Syndromes (Including Cowden Syndrome) | RAD51 | |
| Hereditary Diffuse Gastric Cancer (HDGC) | SMARCA4 | |
| Peutz-Jeghers Syndrome (PJS) | – | |
| PALB2 | – |
|
| De Novo Pathogenic Variant Rate | – |
As we learn more about the genomic changes predisposing a person to cancer, we have been able to make screening tests available to many more people. The specific DNA sequences of the BRCA1 and BRCA2 genes were even the subject of a legal case that went all the way to the United States Supreme Court, who ruled that the sequences of your genes could not be patented. Before this ruling, only one company could provide BRCA1 or BRCA2 testing in the United States, but now there are a number of companies who can help if you’d like to have genomic testing for hereditary causes of breast cancer.
Tom explains what genetic testing means to him and how he benefited from the information it provided.
Watch Tom’s story: Genomic testing and treatment for Lynch syndrome (3 mins) on YouTube.
Video source: Genomics Education Programme. (2024, February 28). Tom’s story: genomic testing and treatment for Lynch syndrome [Video]. YouTube. https://www.youtube.com/watch?v=RwKH0SU-E2g
Appropriate Referrals
Oncology nurses are often a primary point of contact for oncology patients. Oncology nurses collect comprehensive health histories including family histories that may include findings indicative of possible hereditary cancer predisposition. The Oncology Nursing Society (ONS) has created this excellent handout Why it is Important to know if Your Patient’s Cancer is Hereditary, which details the hereditary and red flag indicators that the patient would benefit from further consultation for genetic testing (click the link then click “download .pdf). Oncology nurses should become familiar with the protocols in their jurisdiction about how to initiate referrals for further consultation or genetic testing when they find health assessment data suggestive of a possible hereditary cancer predisposition.
Oncology Resources
This unit provided a brief introduction to oncology genomics, as within this practice context genomics is extensively integrated. Nurses working in oncology need more than a foundational level of genomic literacy. There are ample professional development opportunities available for nurses wishing to learn more about this important area of genomics nursing.
Nursing organizations
- The Oncology Nursing Society (ONS) have a wealth of professional development resources for oncology nurse members, including a Genomics and Precision Oncology Learning Library.
- The Canadian Association of Nurses in Oncology also has numerous resources for Canadian oncology nurses, including a Genomics Oncology Nursing Special Interest Group.
Student membership is free for both of these organizations!
- Linkage (LInking Nursing Knowledge And GEnomics) has online learning modules, including one on cancer genomics.
Oncology Genomics
- The National Comprehensive Cancer Network (NCCN) provides an excellent clinical resource Guidelines for Treatment by Cancer Type, and cancer Detection, Prevention, and Risk Reduction.
- My Cancer Genome offers genetically-informed cancer medicine resources including current information on variants, therapeutic implications, and clinical trials.
- The National Cancer Institute has a cancer genetics overview for healthcare professionals.
Attribution & References
Except where otherwise noted, this page is adapted from Cancer Genomics by Genomics Education Programme, CC BY-NC 4.0 and Cancer Genomics courtesy: National Human Genome Research Institute, Public Domain with attribution
- Proto-oncogenes & Tumour suppressant genes sections adapted from Section 13.5: Oncogenes In Introduction to Genetics , CC BY-SA 4.0 license and was authored, remixed, and/or curated by Ying Liu. / A derivative of Online Open Genetics (Nickle and Barrette-Ng) by Todd Nickle and Isabelle Barrette-Ng is licensed CC BY-SA 3.0 and source content that was edited to the style and standards of the LibreTexts platform.
- Is cancer hereditary? adapted from The Genetics of Cancer by National Cancer Institute (NIH), Public Domain with attribution
- Blood Tests to Detect Cancer adapted from Cancer Genomics courtesy: National Human Genome Research Institute, Public Domain with attribution
References
National Cancer Institute (NCI). (2024, November 22). Genetics of breast and gynecologic cancers (PDQ®) – Health professional version. https://www.cancer.gov/types/breast/hp/breast-ovarian-genetics-pdq#_156

