7.2 Application of Theory – Readings & Case Studies
Optional Reading
This article uses a case study of a family with a history of breast and prostate cancer to highlight the importance of coordinated genetic care by genetics professionals. The article argues that fragmented genetic care can lead to errors such as inappropriate testing, miscommunication of results, and missed opportunities for cancer prevention and early detection, ultimately resulting in psychosocial distress and increased healthcare costs.
Read
Mahon S. M. (2019). Coordination of genetic care: More important and complicated than it seems. Journal of the National Comprehensive Cancer Network, 17(11), 1272–1276. https://doi.org/10.6004/jnccn.2019.7343
Case Study – Clinical Application
Mitochondrial DNA pathogenic variant A1555G and aminoglycoside-induced hearing loss and deafness Case study – a mitochondrial DNA variant causes susceptibility to hearing loss on administration of aminoglycosides.
Key Takeaway:
Carriers of the variant who undergo even one course of aminoglycoside antibiotic therapy can suffer severe an irreversible loss of hearing.
Clinical Scenario:
A term newborn is noted to have an elevated temperature (39 C) and an elevated respiratory rate (45/minute). Sepsis is suspected, blood cultures and laboratory studies are obtained and the child is moved to the intermediate care unit for IV antibiotics (Clinical guidelines recommend Ampicillin and Gentamicin for rule out sepsis). When the mother is informed of the need to start antibiotics she tells the care team that she has hearing loss that she says occurred after receiving an antibiotic that she doesn’t remember. “Some kind of mycin I was told,” She is very concerned that this could happen to her child and asks that the baby be ‘checked out’.
A quick PubMed search using the terms ‘antibiotics’ and ‘hearing loss’ identifies many articles that discuss the risk of hearing loss in individuals exposed to aminoglycoside antibiotics that have a specific mitochondrial pathogenic variant. Given this information the decision is made to start the baby empirically on Ampicillin and a cephalosporin and pursue investigation of the mitochondrial variant.
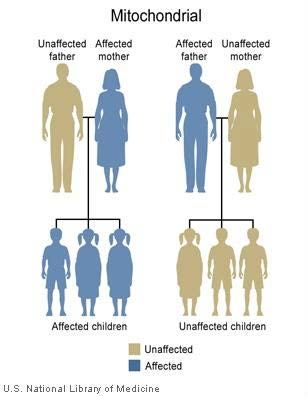
Description of relevant genomic information and how this information would be used:
Mitochondria undergo a special type of inheritance called maternal inheritance. Only the mother contributes mitochondria to her children. Thus, when a mitochondrial DNA variant occurs in one of the maternal mitochondrial genes, she will pass it to all of her offspring. Males do not pass mitochondria to any of their offspring. Mitochondria are involved in the intermediate metabolism of many ingested substances and drugs. Variants in two mitochondrial genes, MT-RNR1 and MT-TS1, confer susceptibility to non-syndromic mitochondrial hearing loss or deafness after treatment with aminoglycoside antibiotics (e.g. gentamicin, kanamycin, streptomycin). Specifically a change from alanine to glycine in position 1555 (“A1555G as well as late onset non-syndromic) sensorineural hearing loss. There are population differences in the prevalence of the A1555G variant: 2.9% – 5.3% in Asian, 0.6% – 2.5 % of Caucasian and as high as 17% of the Spanish population with nonsyndromic hearing loss. Therefore, Asian and Spanish populations have the highest frequency of the A1555G variant followed by other populations of European ancestry. A higher frequency of the variant is found among the deaf population with a history of aminoglycoside exposure accounting for 15-30%. Both males and females are affected equally. The hearing loss is generally bilateral and in the moderate to profound range. Once exposed to aminoglycoside antibiotics, most individuals with the variant go on to develop hearing loss or deafness (Usami & Nishio, 2018; Rehm et al., n.d.; Vivero et al., 2012; Xing et al., 2007).
Recommended clinical action: Genetic testing for the A1555G variant should be performed in individuals with moderate to profound hearing loss in the presence of either a family history of hearing loss suggestive of maternal inheritance or onset of hearing loss following administration of an aminoglycoside antibiotic such as gentamicin. For women who carry the A1555G variant, with or without hearing loss, carrier testing is recommended for other maternal family members with instructions for their children and all other maternal members to strictly avoid the administration of aminoglycoside antibiotics if they carry the variant. Carriers of the variant who undergo even one course of aminoglycoside antibiotic therapy can suffer severe and irreversible loss of hearing. As a cautionary note, lack of identification of the variant does not rule out hearing loss attributable to other variants within mitochondrial genes (e.g. the MT-RNR1 gene) or due to other genes known to be involved in hearing loss (Guan et al., 2006).
Family Implications: Hearing loss caused by this pathogenic variant (A1555G) is consistent with a maternal pattern of inheritance.
Evidence to support the use of genomic information in this scenario: ACMG Practice Guideline: Genetics Evaluation Guidelines for the Etiologic Diagnosis of Congenital Hearing Loss
Source: Case study: Mitochondrial DNA mutation A1555G and aminoglycoside-induced hearing loss and deafness [PDF] from Family Health History for Healthcare Professionals Courtesy: National Human Genome Research Institute (NHGRI), Public Domain with attribution .
Resources:
ACMG (2002). Genetics Evaluation Guidelines for the Etiologic Diagnosis of Congenital Hearing Loss. Genetic Evaluation of Congenital Hearing Loss Expert Panel. ACMG statement. Genetics in medicine : official journal of the American College of Medical Genetics, 4(3), 162–171. https://doi.org/10.1097/00125817-200205000-00011
US Preventive Services Task Force (2008). Universal screening for hearing loss in newborns: US Preventive Services Task Force recommendation statement. Pediatrics, 122(1), 143–148. https://doi.org/10.1542/peds.2007-2210
NIH. (2023, September 28). Genetic hearing loss overview. GeneReviews. https://www.ncbi.nlm.nih.gov/books/NBK1434/
PharmGKB. (n.d.). MT-RNR1 m.1555A>G. https://www.pharmgkb.org/haplotype/PA166229255
Medline Plus. (2014, May 1). MT-TS1 gene. https://medlineplus.gov/genetics/gene/mt-ts1/
Attribution & References
- Case study: Mitochondrial DNA mutation A1555G and aminoglycoside-induced hearing loss and deafness [PDF] from Family Health History for Healthcare Professionals courtesy: National Human Genome Research Institute (NHGRI), Public Domain with attribution . / References changed to APA format.
References
Guan, M., Yan, Q., Li, X., Bykhovskaya, Y., Gallo-Teran, J., Hajek, P., Umeda, N., Zhao, H., Garrido, G., Mengesha, E., Suzuki, T., Del Castillo, I., Peters, J. L., Li, R., Qian, Y., Wang, X., Ballana, E., Shohat, M., Lu, J., . . . Fischel-Ghodsian, N. (2006). Mutation in TRMU Related to Transfer RNA Modification Modulates the Phenotypic Expression of the Deafness-Associated Mitochondrial 12S Ribosomal RNA Mutations. The American Journal of Human Genetics, 79(2), 291–302. https://doi.org/10.1086/506389
Rehm, H. L., Williamson, R. E., Kenna, M. A., Corey, D. P., Korf, B. R., & Harvard Medical School Center For Hereditary Deafness. (n.d.). Understanding the genetics of deafness. In Harvard Medical School Center for Hereditary Deafness. https://projects.iq.harvard.edu/files/centerforhereditarydeafness/files/understanding_the_genetics_of_deafness.pdf
Usami S, & Nishio S. (2018, June 14). Nonsyndromic hearing loss and deafness, Mitochondrial. In Adam MP, Feldman J, Mirzaa GM, et al., (Eds). GeneReviews®. University of Washington. https://www.ncbi.nlm.nih.gov/books/NBK1422/
Vivero, R. J., Ouyang, X., Yan, D., Du, L., Liu, W., Angeli, S. I., & Liu, X. Z. (2012). Mitochondrial DNA mutation screening in an ethnically diverse nonsyndromic deafness cohort. Genetic testing and molecular biomarkers, 16(9), 1146–1148. https://doi.org/10.1089/gtmb.2011.0365
Xing, G., Chen, Z., & Cao, X. (2007). Mitochondrial rRNA and tRNA and hearing function. Cell Research, 17(3), 227–239. https://doi.org/10.1038/sj.cr.7310124


