15 Introduction
Chapter Objectives
- Early Ideas in Atomic Theory
- Evolution of Atomic Theory
- Atomic Structure and Symbolism
- Chemical Formulas
- The Periodic Table
- Molecular and Ionic Compounds
- Chemical Nomenclature
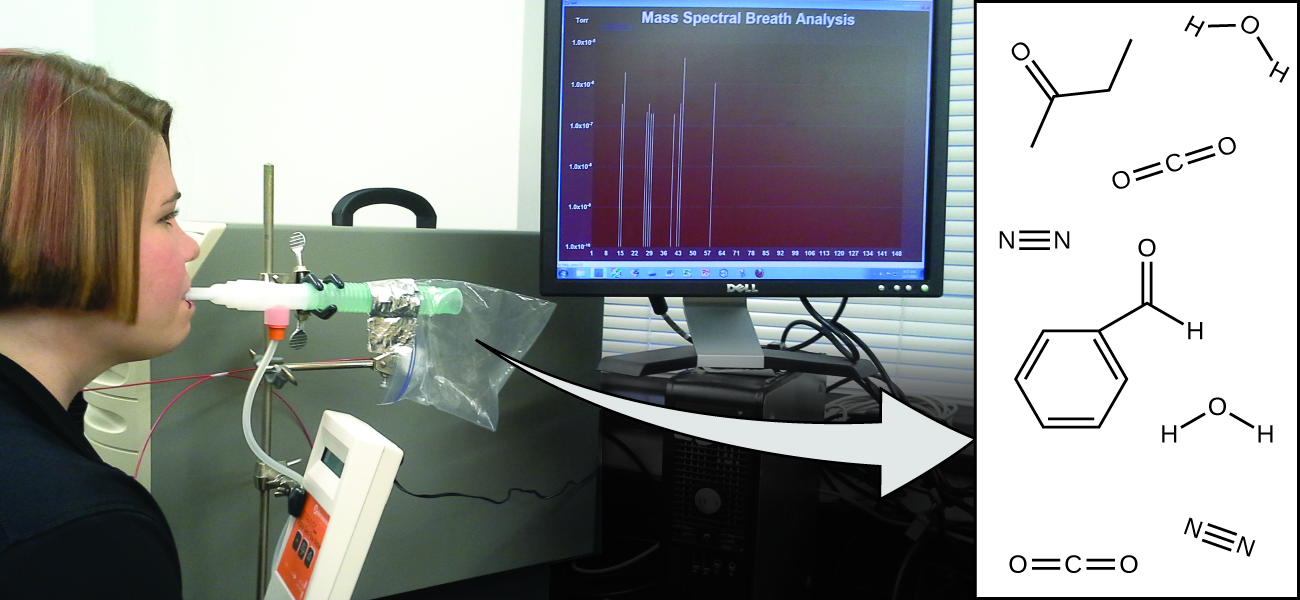
Your overall health and susceptibility to disease depends upon the complex interaction between your genetic makeup and environmental exposure, with the outcome difficult to predict. Early detection of biomarkers, substances that indicate an organism’s disease or physiological state, could allow diagnosis and treatment before a condition becomes serious or irreversible. Recent studies have shown that your exhaled breath can contain molecules that may be biomarkers for recent exposure to environmental contaminants or for pathological conditions ranging from asthma to lung cancer. Scientists are working to develop biomarker “fingerprints” that could be used to diagnose a specific disease based on the amounts and identities of certain molecules in a patient’s exhaled breath. An essential concept underlying this goal is that of a molecule’s identity, which is determined by the numbers and types of atoms it contains, and how they are bonded together. This chapter will describe some of the fundamental chemical principles related to the composition of matter, including those central to the concept of molecular identity.